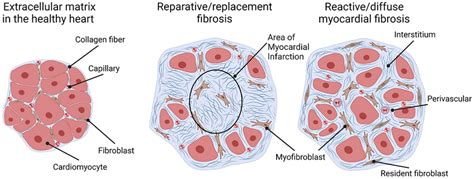
Fibrosis is a pathological condition characterized by the excessive accumulation of components that form the tissue’s architecture, known as the extracellular matrix (ECM). This buildup leads to increased tissue stiffness and can ultimately impair organ function.
The Role of Extracellular Matrix and Tissue Stiffness
The extracellular matrix (ECM) provides structural support to tissues. In healthy organs, cells meticulously regulate the deposition of ECM components, such as collagen, to maintain tissue integrity. However, during fibrotic processes, an imbalance occurs, leading to increased ECM deposition and subsequent tissue stiffening.
It is widely thought that fibrosis occurs when components that hold together a tissue’s architecture (called the extracellular matrix (ECM)) build up in the tissue and lead to tissue stiffness. “We knew that stiffness is an important factor in the build-up of scar tissue in the lung,” explains lead author Mark Jones, NIHR Clinical Lecturer in Respiratory Medicine at the NIHR Southampton Biomedical Research Centre and University of Southampton, UK. “But we didn’t understand what specifically causes increased stiffness in diseased human tissue.”
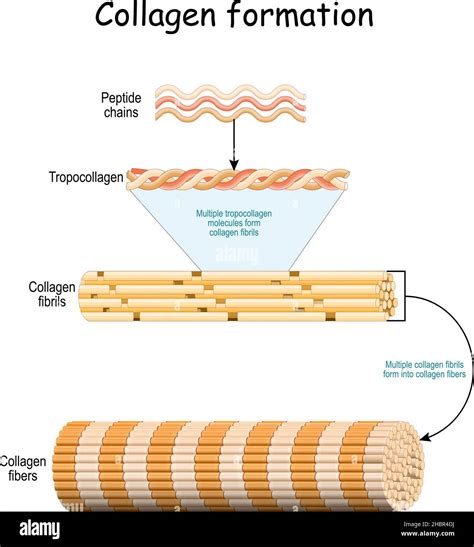
Previous studies have indicated that fibrosis is initiated by changes in the mechanical properties of collagen resulting from modifications to its structure. Experiments have found that, after tissue injury, a family of enzymes called lysyl oxidases (LOXs) increase the bonds between individual collagen fibers in the organ. This altered structure makes the tissue stiffer, driving the fibrotic process.
When our bodies heal following an injury, fibroblasts and other cells deposit components of the extracellular matrix, such as collagen, that form a ‘scar’ over the damaged area. In healthy organs, fibroblasts lay down just enough collagen to maintain the integrity of the tissue. However, if the tissue begins to stiffen, these cells produce more collagen, disrupting this equilibrium. This increases the rigidity of the organ, triggering the fibroblasts to release even more collagen.
Investigating Collagen Structure in Fibrosis
Researchers began by examining the biological and mechanical features of lung tissue from individuals with lung fibrosis and compared these to healthy lung tissue. A key focus was on enzymes that impart collagen its unique ‘cross-linked’ structure within the ECM. They discovered that a specific family of these enzymes, known as the LOXL family, was more abundant in fibrotic samples.
This observation led to a deeper investigation into the types of collagen structures present in fibrotic samples. These structures are broadly categorized into immature and mature collagen cross-links. The findings revealed that increased lung tissue stiffness was specifically associated with higher amounts of mature cross-linked collagen. Furthermore, in these samples, the structure of each individual collagen building block, or fibril, was altered.
Targeting Collagen Modification to Prevent Fibrosis
Building on these discoveries, the team explored whether blocking LOXL enzymes could alter collagen structure and potentially prevent lung fibrosis. They tested a compound named PXS-S2A, which inhibits LOXL-2 and LOXL-3, in lung tissue cells isolated from patients with fibrosis.
“Together, our results identify that treatment approaches which maintain normal collagen architecture may prevent tissue stiffening and limit the mechanisms that drive progressive fibrosis,” states Donna Davies, Professor of Respiratory Cell and Molecular Biology at the University of Southampton.
Fibulin-5 and Skin Fibrosis
In parallel research, an international team identified a novel molecule involved in skin fibrosis, a serious condition characterized by inflammation and hardening of the skin. This study revealed a new mechanism for fibrosis development in skin tissues, which could also be relevant to fibrosis in other organs such as the lungs, heart, liver, and kidneys.
The research, led by bioengineering professor Shyni Varghese at the University of California, San Diego, and Colin Jamora, a biologist at the IFOM-inSTEM Joint Research Laboratory in India, highlighted the role of a molecule called fibulin-5 in the development of skin fibrosis in mice.
Fibulin-5 is crucial for the formation of elastin, a protein that allows skin to regain its original shape after stretching. While much research on fibrosis causes has focused on collagen overproduction, this study shifted focus to elastin. The team discovered that fibrosis development in skin tissues was linked to fibulin-5 levels.
Researchers observed substantially higher levels of fibulin-5 in the skin tissues of mice genetically engineered to develop skin fibrosis compared to normal mice. Elevated fibulin-5 levels were also detected in the skin tissues of human patients with skin fibrosis. The next steps involve exploring methods to inhibit fibulin-5 increase and determine its role in fibrosis in other organs.
The Role of Hypoxia-Inducible Factors (HIFs)
Further investigations into idiopathic pulmonary fibrosis (IPF), a disease causing severe lung scarring of unknown cause, revealed the involvement of Hypoxia Inducible Factors (HIFs). These proteins regulate the body’s response to varying oxygen levels.
Researchers extracted fibroblasts from IPF patients and cultured them in the laboratory. They found that HIFs activated the genes for enzymes PLOD2 and LOXL2, which are involved in modifying collagen. This activation increased the number of cross-links between collagen fibers, leading to increased stiffness. Electron microscopy revealed that the collagen fibers in these cells had a smaller diameter, consistent with observations in IPF patients.
HIF activity is regulated by a protein called FIH (Factor Inhibiting HIF). FIH is inactivated by oxidative stress, leading to a state called ‘pseudohypoxia,’ where cells behave as if oxygen is low even under normal conditions. These findings suggest that pseudohypoxia, induced by oxidative stress, is central to IPF pathology. Injury or damage to lung tissue can increase oxidative stress, reducing FIH levels. This allows HIF to activate PLOD2 and LOXL2, altering collagen structure and stiffening the tissue, thereby initiating the fibrosis cycle.
Collagen Diversity and Fibrosis Progression
The extracellular matrix in fibrotic tissues is often described as dense collagen. However, fibrosis is not a monolithic condition, as evidenced by variations in its presentation across different organ injuries (e.g., viral, cholestatic, autoimmune, or toxic liver injury). The composition of the ECM, particularly the diverse types of collagens, significantly influences cellular functions and physiological processes, changing dramatically during fibrosis progression.
It is hypothesized that different types of collagens can be beneficial or detrimental in fibrosis, with their function potentially changing based on their location. For instance, basement membrane collagen type IV anchors cells in a polarized manner, while interstitial collagens like type I and III do not provide directional information. Furthermore, biologically active degradation products of certain collagens can influence cell function, proliferation, and matrix production. Examples include the carboxyterminal propeptides of interstitial collagen type VI (endotrophin) and basement membrane collagen type XVIII (endostatin), which act as regulators of angiogenesis.
A comprehensive understanding of the 28 known collagen types and their precise roles is crucial. The molecular composition of the ECM in fibrosis warrants careful attention to accurately assess its impact on organ function and its potential for progression or reversal.