The recent FDA approval of new oral medications offers effective therapies for individuals experiencing severe hair loss due to alopecia areata. This autoimmune condition, which causes hair to fall out in clumps, significantly impacts patients' lives, often beginning in their 30s, a critical period for personal and professional development.
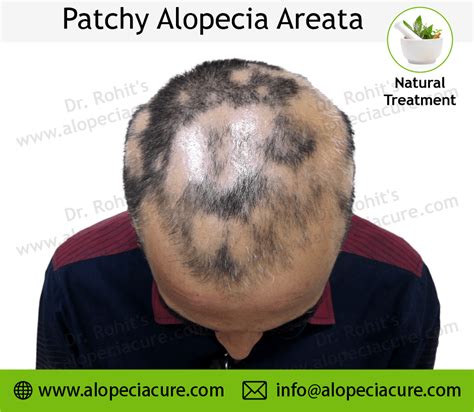
Understanding Alopecia Areata
Alopecia areata is an autoimmune disease where the immune system mistakenly attacks hair follicles, leading to sudden, patchy hair loss. It's important to note that alopecia areata does not typically cause total baldness, as it does not affect the entire scalp. However, hair loss affecting the entire scalp is known as alopecia totalis, and when the entire body and scalp lack hair, it is termed alopecia universalis. In both these more extensive forms, the immune system is also considered the primary cause.
New FDA-Approved Treatments
OLUMIANT (baricitinib)
OLUMIANT (baricitinib) represents a groundbreaking new drug approved by the FDA for adults with alopecia areata. As the first systemic drug for this condition, OLUMIANT treats the entire body rather than targeting only the site of hair loss. It functions as a JAK inhibitor, interrupting the activity of specific enzymes involved in the inflammatory response believed to cause alopecia areata.
Dr. Mesinkovska described OLUMIANT as "a godsend for patients suffering from Alopecia Areata." Previously, steroid injections into the lesions were the primary treatment. Now, relief from patchy hair loss across the scalp and body can be achieved with a pill.
Clinical Trials and Efficacy
Before its FDA approval, OLUMIANT was tested in two double-blind, placebo-controlled, randomized trials lasting 36 weeks. Participants had severe scalp hair loss (at least 50 percent) for six months or longer and received either a placebo or OLUMIANT daily. In the first trial, 57 percent of subjects taking OLUMIANT achieved significant scalp hair coverage (80 percent), compared to only five percent in the placebo group. The second trial demonstrated similar positive outcomes. Improvements in eyebrow and eyelash coverage were also observed in participants experiencing hair loss in these areas.
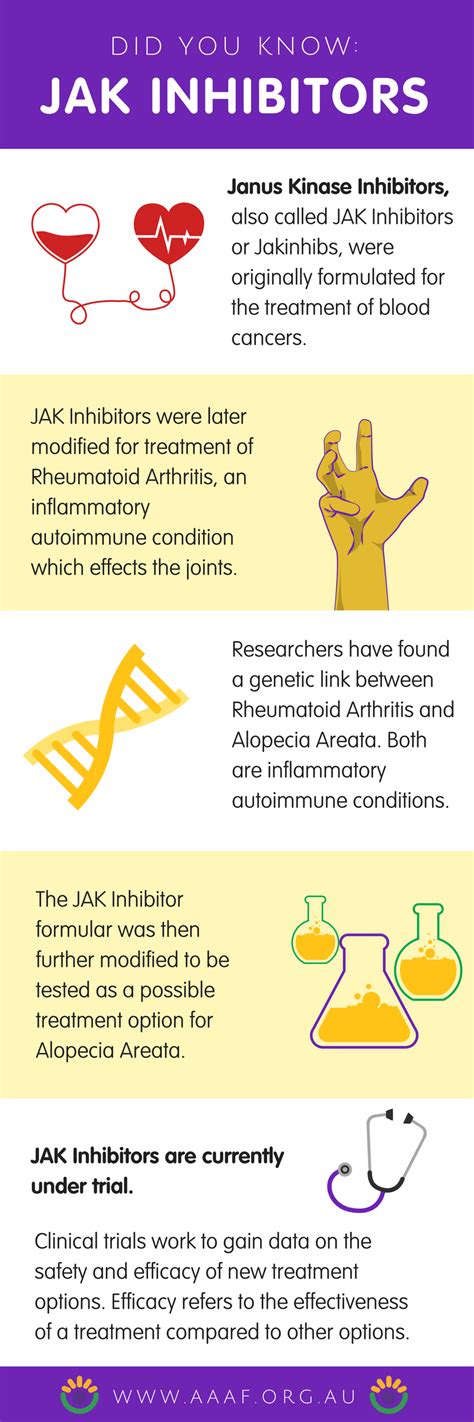
Potential Side Effects of OLUMIANT
The most common side effects associated with OLUMIANT include headache, nausea, acne, fatigue, urinary tract infections (UTI), shingles, elevated cholesterol, respiratory infections, changes in liver enzymes, folliculitis, anemia, yeast infections, reduced white blood cell count, abdominal pain, and weight gain. More serious potential risks include severe infections, mortality, cardiovascular issues, and thrombosis. Therefore, careful patient screening and monitoring are essential.
OLUMIANT is an immunosuppressant and may not be suitable for everyone. It is not recommended for individuals taking other JAK inhibitors, cyclosporine, biologic immunomodulators, or certain other immunosuppressants. Doctors may need to monitor patients using OLUMIANT closely.
Ritlecitinib (Litfulo)
On June 23, the FDA announced its approval of ritlecitinib, another Janus kinase (JAK) inhibitor, for the treatment of alopecia areata in both adolescents and adults. Dr. Brett King, who was involved in the clinical trials with ritlecitinib, stated that this approval "changes the treatment landscape for people with alopecia areata enormously."
The approval of ritlecitinib is particularly significant as alopecia areata commonly affects children and adolescents, vulnerable periods where hair loss can profoundly impact their social development and emotional well-being, leading to withdrawal, anxiety, and sadness.
Alopecia Areata in Children and Adolescents: More than Just Skin Deep
Other Treatment Options for Alopecia Areata
While there is currently no cure for alopecia areata, several treatments can help regrow hair in both adults and children by blocking the immune system's attack on hair follicles. It is important to note that not all treatments work for everyone, and finding the right one may require time and patience.
Corticosteroids
Corticosteroids, or steroids, work by reducing inflammation around hair follicles, which can promote hair growth. They are often used in combination with other alopecia areata treatments.
- Intralesional injections: For adults with patchy alopecia areata, intralesional corticosteroid injections into the bald patches on the scalp or face are a common treatment. While some individuals experience pain, others report only slight discomfort. New hair growth may be observed within six to eight weeks, and treatments can be repeated every four to six weeks. However, these injections do not prevent further hair loss.
- Oral steroids: Pulse corticosteroids involve high doses given at regular intervals over a short period. While they can be effective, long-term use of oral steroids is not recommended due to potential side effects such as weight gain, osteoporosis, elevated blood sugar levels, and high blood pressure.
Minoxidil
Oral Minoxidil is available in tablet form at strengths of 2.5 mg, 5 mg, or 10 mg. For patients with moderate disease, topical corticosteroids combined with topical or oral minoxidil may be considered.
Immunotherapy
This treatment involves applying an agent that causes an allergic rash (allergic contact dermatitis) at the application site. While the exact mechanism is not fully understood, this process is believed to alter the immune response around hair follicles, promoting regrowth. The rash can resemble poison oak or ivy and may cause redness and discomfort. Reports suggest significant regrowth in 40 to 55 percent of patients, but continuous treatment is often necessary to maintain results.
Dupilumab (Dupixent)
Approximately one-third of individuals with alopecia areata also have atopic dermatitis or eczema. Dupilumab (Dupixent) is a monoclonal antibody FDA-approved for treating atopic dermatitis in adults and children as young as six months.
Treatments Not Recommended for Alopecia Areata
Hair transplant surgery is generally not advised for treating alopecia areata. This is because hair transplant candidates require a sufficient supply of healthy donor hair follicles, which individuals with severe alopecia often lack.

The Future of Alopecia Areata Treatment
While significant progress has been made with the recent FDA approvals of baricitinib and ritlecitinib, research continues. Dr. King highlighted the ongoing need for approvals for younger patients and the development of treatments beyond JAK inhibitors, as they do not work for everyone. The ultimate goal is to ensure effective treatment options are available for all individuals affected by alopecia areata.