Tretinoin, also known as all-trans retinoic acid (ATRA), is an endogenous metabolite of vitamin A that is naturally present in plasma. While sporadic case reports have suggested a link between maternal use of topical tretinoin during the first trimester of pregnancy and malformations, including cardiovascular defects, limb defects, ear defects, and CNS defects, larger cohort studies have not demonstrated an increased risk of congenital malformation following first-trimester topical tretinoin exposure.
Currently, there is no substantial evidence indicating that topical tretinoin exposure is associated with an increased risk of miscarriage, low birth weight, preterm delivery, or intrauterine death. However, the available data are considered too limited to definitively rule out fetal risk, and therefore, its use during pregnancy is generally not recommended.
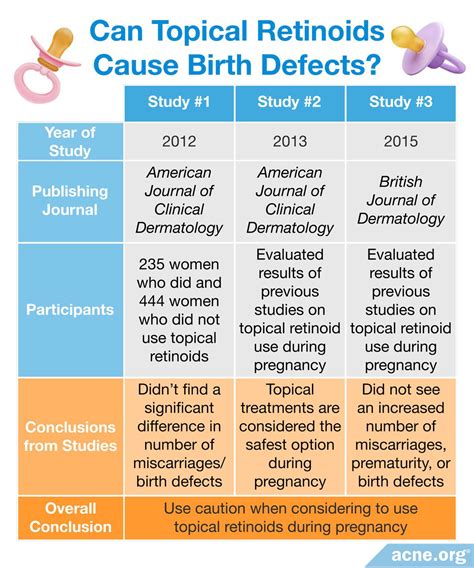
Manufacturer Recommendations and Oral Tretinoin Use
The manufacturer advises a high risk of severe malformations associated with tretinoin. For oral treatment, it is mandated that effective contraception must be used throughout the duration of treatment and for one month afterward. Progesterone-only pills are not considered an effective contraceptive measure during tretinoin treatment.
There is insufficient data, particularly concerning first-trimester exposure, to quantify the risks posed to a developing fetus following oral exposure to tretinoin. Case reports have described miscarriage, intrauterine fetal death, low birth weight, and preterm delivery after oral tretinoin exposure; however, the impact of the underlying condition cannot be excluded in these instances.
The risk-benefit balance between maternal and fetal well-being must be assessed on an individual basis. Other retinoids are known teratogens at therapeutic doses, and the potential for an increased risk of structural malformation and neurodevelopmental impairment with tretinoin use in the first trimester should be considered and discussed with the patient.
Risks Associated with Other Retinoids
Data regarding the risk of adverse neurodevelopmental effects following in utero tretinoin exposure are limited, but adverse effects are well-documented for other retinoids. For instance, in rats and rabbit studies, topical tazarotene has caused reduced fetal body weights, reduced skeletal ossification, and retinoid malformations such as spina bifida, hydrocephaly, and cardiovascular anomalies. Although 8 out of 9 reported human pregnancies inadvertently exposed to topical tazarotene delivered apparently healthy babies, tazarotene is classified as pregnancy category X and should be avoided during pregnancy due to its retinoid nature and reported adverse effects in animal studies.
Topical isotretinoin is absolutely contraindicated in pregnancy due to extensive data showing increased pregnancy loss in the first trimester and numerous birth defects, including hydrocephalus, cleft palate, ear canal stenosis, thymic defects, spina bifida, and cardiac conotruncal malformations. In the United States, isotretinoin users are required to register with the iPLEDGE program, a national registry designed to prevent fetal exposure. Periodic pregnancy tests are mandatory before drug refills are issued.

Topical Tretinoin: Understanding the Risks
Topical tretinoin is a medication applied to the skin to treat acne, prevent wrinkles, and address other skin concerns. It belongs to the retinoid class of medications, which are related to Vitamin A. Other retinoids include isotretinoin, acitretin, and adapalene.
While tretinoin is related to isotretinoin, a known teratogen, it is taken orally and enters the bloodstream at higher levels compared to topical tretinoin. The skin generally acts as a good barrier, limiting the amount of tretinoin absorbed into the bloodstream with topical use as directed. Higher absorption can occur if tretinoin is used on broken or irritated skin, in excessive amounts, or over a large body area. Generally, less topical tretinoin used on the skin correlates with lower fetal risk.
Several studies have investigated the safety of topical tretinoin in pregnancy. These studies have not identified a higher chance of birth defects with proper application. However, case reports have described birth defects in babies born to women who used topical tretinoin during pregnancy. These reported birth defects are similar to those observed with oral isotretinoin exposure, suggesting a potential for similar effects due to the relationship between the two medications. Despite these reports, the chance of birth defects is likely low, as many individuals have used tretinoin during pregnancy without adverse outcomes.
Tretinoin Use During Different Trimesters
During the first trimester, a baby's organs are forming. Tretinoin use in the second and third trimesters is less likely to cause a birth defect, as the baby's body and brain are primarily growing during these periods. However, until more information is available, avoiding topical tretinoin throughout pregnancy may be the safest approach.
Tretinoin and Breastfeeding
The safety of tretinoin use during breastfeeding has not been extensively studied. However, given that very little tretinoin is absorbed into the body with topical application, the amount that may pass into breast milk is likely to be small.
Tretinoin and Male Fertility
Studies have not been conducted to determine if topical tretinoin use could affect male fertility or increase the chance of birth defects. Generally, exposures by fathers or sperm donors are considered unlikely to increase risks to a pregnancy.
Nordic Cohort Study Findings
A recent study analyzing nationwide health and prescription registries across Denmark, Iceland, Norway, and Sweden examined topical retinoid use among women of reproductive age. The study found a significant increase in topical retinoid use, more than tripling between 2006 and 2024. Evaluating pregnancy exposures from nearly 3.87 million births between 1996 and 2020, researchers found that only a small proportion of infants (0.86 per 1,000) had confirmed first-trimester exposure to topical retinoids. Among these exposed infants, 3.3% were diagnosed with a major congenital malformation, compared to 3.0% in unexposed infants. After adjusting for confounding factors, this difference was not statistically significant. While these findings are reassuring, the authors caution that focusing only on live births might underestimate both the frequency of topical retinoid use in early pregnancy and the true incidence of malformations.
Tips for Skincare and Retinol While Trying to Conceive and in Early Pregnancy - Dr Lora Shahine
Oral Isotretinoin: Severe Risks
Oral isotretinoin is prescribed for severe nodular acne that has not responded to other treatments and can cause lasting scars. It is a potent oral retinoid that can lead to serious problems for a baby during pregnancy, including:
- Miscarriage
- Preterm birth (before 37 weeks of pregnancy)
- Birth defects (changes in the shape or function of body parts)
- Intellectual and developmental disabilities later in life
- Cleft palate and other facial birth defects
- Congenital heart defects
- Ear problems (small or missing ears)
- Eye problems (e.g., microphthalmia, leading to vision loss)
- Microcephaly (baby's head smaller than expected)
- Problems with the thymus and parathyroid glands
Due to the severe teratogenicity of oral isotretinoin, strict protocols, including the iPLEDGE program in the US, are in place to prevent fetal exposure. This program requires registration and adherence to strict rules regarding medication use and contraception. It is crucial for individuals taking isotretinoin to use two effective forms of birth control and to inform their healthcare provider about their contraceptive methods, as some birth control pills may be less effective when taken with isotretinoin.
Men taking isotretinoin may have small amounts in their semen. While experts are unsure if this can lead to birth defects, pregnant partners or those planning pregnancy should consult their provider. Donating blood while taking isotretinoin or for one month after the last dose is also prohibited.
Other Oral Retinoids and Pregnancy
Other oral retinoids, such as acitretin (Soriatane®) for severe psoriasis, tretinoin (Vesanoid®) for a type of blood cancer, and bexarotene (Targretin®) for a rare skin cancer, can also cause birth defects similar to isotretinoin. Although there isn't a specific program like iPLEDGE for these medications, following similar precautionary measures can help prevent birth defects.
Vitamin A Intake During Pregnancy
Retinoids are man-made forms of vitamin A, which is essential for healthy fetal growth and development. While the body can produce its own vitamin A from certain yellow and green vegetables, which is safe during pregnancy, excessive intake of preformed vitamin A, found in foods like meat (especially liver), fish, and dairy, can cause birth defects. It is important to discuss vitamin A intake with a healthcare provider to ensure adequate yet safe levels during pregnancy.