Psoriasis is a common, long-term (chronic) disease that affects approximately 2-3% of the global population. While there is no known cure, effective treatments are available to manage its symptoms. The condition is characterized by cycles of flares, which can last for weeks or months, followed by periods of remission.
Understanding Psoriasis
Psoriasis is thought to stem from an immune system problem where infection-fighting cells mistakenly attack healthy skin cells, causing them to grow much faster than usual. Researchers believe that both genetic predisposition and environmental factors play significant roles in its development. Many individuals genetically predisposed to psoriasis may remain symptom-free for years until a specific environmental factor triggers the disease.
While anyone can develop psoriasis, about a third of cases begin in childhood. Having a parent with psoriasis increases an individual's risk. Smoking is also identified as a risk factor.
Types of Psoriasis
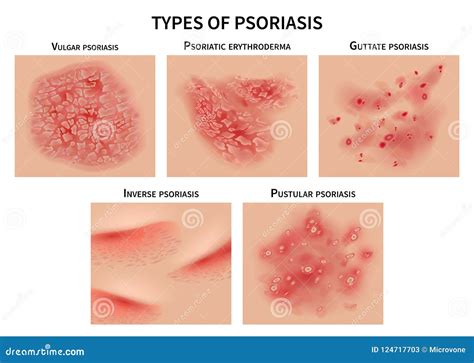
Psoriasis can manifest in several forms, each with distinct characteristics:
Plaque Psoriasis
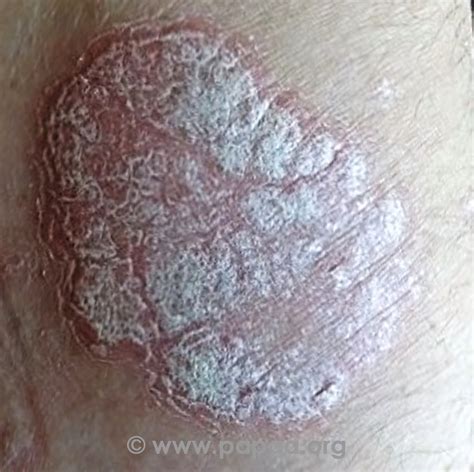
This is the most common type, characterized by dry, itchy, raised skin patches known as plaques, often covered with scales. These plaques can vary in number and appearance, depending on skin color, and commonly appear on the elbows, knees, lower back, and scalp.
Nail Psoriasis
Psoriasis can affect both fingernails and toenails, leading to changes such as pitting, abnormal growth, and discoloration. A common symptom is onycholysis, where the nail loosens and separates from the nail bed.
Guttate Psoriasis
Primarily affecting young adults and children, guttate psoriasis is often triggered by a bacterial infection, such as strep throat. It typically presents as small, drop-like lesions.
Inverse Psoriasis
This type primarily affects the skin folds in areas like the groin, buttocks, and under the breasts. It appears as smooth, inflamed patches that can worsen with friction and sweating.
Pustular Psoriasis
A rarer form, pustular psoriasis is characterized by clearly defined, pus-filled blisters.
Erythrodermic Psoriasis
The least common type, erythrodermic psoriasis can cause a widespread, peeling rash that covers the entire body and may be accompanied by intense itching or burning sensations.
Genetic Basis of Psoriasis
Genetic factors are crucial in the etiology of psoriasis. The condition has a higher prevalence among relatives of affected individuals, and concordance rates are higher in monozygotic twins compared to dizygotic twins. Linkage studies have identified at least nine genomic regions (loci) associated with psoriasis, known as PSORS1 through PSORS9.
The PSORS1 region, located within the major histocompatibility complex (MHC), plays a significant role in antigen presentation and includes the corneodesmosin gene, vital for keratinocyte cohesion. This locus accounts for a substantial portion of the disease's heritability.
Other associated loci include PSORS2 on chromosome 17q25, which contains the CARD14 gene involved in nuclear factor-κB (NF-κB) activation, and PSORS4 on chromosome 1q21, linked to genes involved in the terminal differentiation of the stratum corneum. Genome-wide association studies (GWAS) have further pinpointed these regions and identified additional susceptibility loci by analyzing genetic markers across the genome in large populations.
GWAS, while powerful, primarily reveal statistical associations. The identified genetic variants may serve as markers for causal variants due to linkage disequilibrium. International collaborations and meta-analyses of GWAS data are essential for increasing statistical power and discovering novel loci.
Pathogenic Pathways
The development of psoriatic inflammation involves both innate and adaptive immune responses. In chronic plaque psoriasis, innate responses are particularly significant. A key mechanism involves the overexpression of antimicrobial peptides, such as LL-37, which act as triggers and contribute to the ongoing maintenance of the condition.
LL-37, released by damaged keratinocytes, forms complexes with other molecules, including genetic material. This complex activates plasmacytoid dendritic cells, stimulating the production of type I interferon (IFN). IFN plays a crucial role in the maturation of myeloid dendritic cells and the differentiation of T-helper 1 (Th1) and T-helper 17 (Th17) cells. Th17 cells produce critical cytokines like IL-17, IL-22, IL-21, and TNF-α.
Cytokines such as IL-6, IL-1β, and IL-23 are vital for activating Th17 cells, which can drive chronic inflammation and autoimmunity. The migration of myeloid dendritic cells to lymph nodes is associated with increased production of TNF-α, IL-23, and IL-12.
The maintenance phase of psoriatic inflammation is sustained by adaptive immune responses, specifically through T cell subsets. Keratinocyte proliferation in the epidermis is driven by pathways involving TNF-α, IL-17, and IFN-γ, as well as the LL-37/DNA complex leading to type I IFN production. These mediators further activate keratinocytes, perpetuating inflammation.
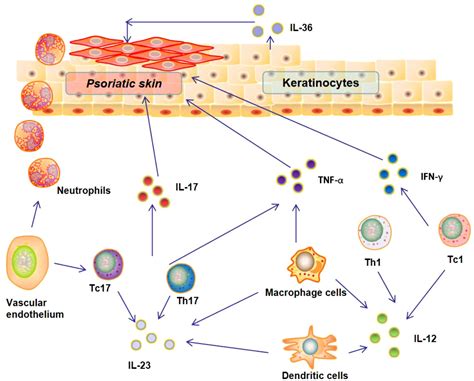
Role of Cytokines
Plaque-type psoriasis is characterized by an inflammatory pathway involving TNF-α, IL-23, and Th17 cells. Various cell types, including CD8+ T cells, invariant NKT cells, γδ T cells, type 3 innate lymphoid cells, and neutrophils, produce IL-17A-F cytokines, which regulate inflammatory responses. The IL-17A/IL-17F receptor signaling pathway is particularly critical. Activation of intracellular kinases leads to the expression of pro-inflammatory cytokines, chemokines, and antimicrobial peptides.
Pustular Psoriasis Pathogenesis
In contrast to plaque psoriasis, where the adaptive immune system plays a major role, the innate immune system appears more central to the pathogenesis of pustular psoriasis. This type of psoriasis is marked by sterile pustules and is often associated with increased expression of IL-1β, IL-36α, and IL-36γ, which promotes neutrophil accumulation in the epidermis. Neutrophil chemokines like CXCL1, CXCL2, and CXCL8 (IL-8) are also significantly present.
Guttate Psoriasis and Streptococcal Triggers
For guttate psoriasis, which often affects children and teenagers, streptococcal superantigens may stimulate T cell proliferation. Molecular mimicry, where bacterial proteins resemble keratin proteins, might also play a role, particularly in individuals with the HLA-Cw6 allele. CD8+ T cell IFN-γ responses can be triggered by specific peptides in these patients.
Drug-Related Psoriasis
Certain medications can trigger the onset or worsening of psoriasis. These drugs can induce psoriatic lesions in individuals without a prior history or predispose them to the condition. Identifying drug-related causes can be challenging due to variable latency periods.
Clinically and histopathologically, drug-related psoriasis can resemble typical psoriasis, presenting in various forms. However, eosinophilic infiltrates and lichenoid reactions in the dermis can sometimes suggest a drug-induced cause. While capillary changes in the upper dermis might differ from conventional psoriasis, these are not definitive indicators.
Commonly implicated drugs include β-blockers, lithium, antimalarial drugs, interferons, and angiotensin-converting enzyme inhibitors. The exact mechanisms are complex, but some drugs affect keratinocyte hyperproliferation and the IL-23/IL-17 signaling pathway. For instance, β-blockers can decrease intraepidermal cAMP, leading to hyperproliferation.
Immune checkpoint inhibitors and molecular inhibitors used in cancer and autoimmune disease treatment can also influence the immune system and potentially trigger psoriasis. Paradoxical reactions to biologics, especially TNF-α inhibitors and those targeting IL-23 and IL-17, can occur, leading to cytokine imbalances and altered immune responses. If drug-related psoriasis is suspected, discontinuing the offending medication and switching to alternatives is recommended.
Associated Conditions
Psoriasis is often associated with other dermatological conditions, including alopecia areata, vitiligo, hidradenitis suppurativa, chronic spontaneous urticaria, bullous dermatoses, and various autoimmune diseases. Psoriatic arthritis, a form of chronic inflammatory arthritis, affects up to 30% of patients with psoriasis, often involving painful joint inflammation and potentially leading to dactylitis (sausage-shaped swelling of fingers and toes).
Ocular manifestations, such as blepharitis, conjunctivitis, and uveitis, can also occur in approximately 10% of patients. Psoriasis can also impact the quality of life, with higher rates of depression, anxiety, and substance abuse observed in affected individuals.
Clinical Presentation and Diagnosis
The diagnosis of psoriasis is typically made based on clinical morphology and the location of lesions. Characteristic histopathological findings include parakeratosis, micro-abscesses, regular elongation of epidermal ridges, and dilated, tortuous capillaries in the dermal papilla.
Key clinical signs include the Koebner phenomenon, where lesions appear at sites of skin injury, and the Auspitz sign, characterized by pinpoint bleeding points upon removal of psoriatic scales.
Psoriasis is often classified into Type I (early onset, positive family history, HLA-Cw6 association) and Type II (later onset, no family history, no HLA-Cw6 association). The disease can present with various morphologies, including plaque, guttate, rupioid, erythrodermic, pustular, and inverse forms.
When to Seek Medical Advice
If you suspect you may have psoriasis, it is important to consult a healthcare provider for accurate diagnosis and management.
tags: #pathophysiology #for #psoriasis