Benzoyl chloride, also known as benzenecarbonyl chloride, is an organochlorine compound with the chemical formula C6H5COCl. It is a colorless, fuming liquid characterized by a pungent odor.
The density of benzoyl chloride is reported as 10.2 lb/gal. Its flash point is 162°F. As a lachrymator, it is irritating to the skin and eyes, and it is corrosive to metals and tissue.
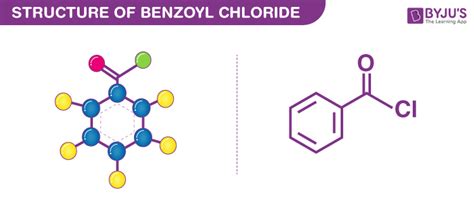
Chemical Properties and Reactivity
Benzoyl chloride is a typical acyl chloride. It reacts with alcohols to form the corresponding esters. Many substituted derivatives of benzoyl chloride are known.
It fumes in air and can decompose exothermically and rapidly in water, forming insoluble benzoic acid and hydrochloric acid (HCl). However, experimental work at Argonne National Laboratory has suggested that under normal conditions, benzoyl chloride is quite unreactive in water, with no HCl formation observed. This does not preclude HCl formation via a catalytic mechanism.
At fire temperatures, benzoyl chloride may react violently with water or steam.
Reactions with Other Substances
Benzoyl chloride reacts violently with protic solvents such as alcohols. It also reacts vigorously with amines and amides, including dimethylformamide. Furthermore, it causes the violent decomposition of dimethyl sulfoxide.
Mixing benzoyl chloride with diisopropyl ether or other ethers in the presence of trace amounts of metal salts may lead to vigorous or explosive reactions.
The Friedel-Crafts acylation of naphthalene using benzoyl chloride, catalyzed by AlCl3, must be conducted above the melting point of the mixture to avoid violent reactions.
Safety and Handling
Benzoyl chloride poses several hazards:
- Inhalation: May irritate the eyes, nose, and throat.
- Ingestion: Causes acute discomfort.
- Skin Contact: Causes irritation and burning.
Firefighting and Emergency Response
Small Fires: Use dry chemical or CO2.
Large Fires: Flood the fire area with large quantities of water, while simultaneously knocking down vapors with water fog.
Fires Involving Tanks, Rail Tank Cars, or Highway Tanks: Cool containers with flooding quantities of water until well after the fire is extinguished. Do not allow water to enter containers. Withdraw immediately if a rising sound is heard from venting safety devices or if the tank shows discoloration. Always stay away from tanks in direct contact with flames.
Spills:
- Stop the leak if it can be done without risk.
- Use water spray to reduce vapors; do not apply water directly to the leak, spill area, or inside containers.
- Small Spills: Cover with dry earth, dry sand, or other non-combustible material. Follow with a plastic sheet to minimize spreading or contact with rain. Use clean, non-sparking tools to collect the material and place it into loosely covered plastic containers for later disposal. Prevent entry into waterways, sewers, basements, or confined areas.
Safe Handling of Chemicals
Personal Protective Equipment (PPE)
Full protective clothing is required, including:
- Full-face respirator for acid gases and organic vapors (yellow GMC canister)
- Close-fitting goggles
- Non-slip rubber gloves
- Plastic apron
- Face shield
Protective clothing should be selected based on the specific conditions of use, considering permeation rates and breakthrough times. Military standard MIL-STD-282 provides guidance for PPE selection.
First Aid Measures
Eyes: First, check for contact lenses and remove them if present. Flush the victim's eyes with water or normal saline solution for 20 to 30 minutes while calling a hospital or poison control center. Do not apply ointments, oils, or medication without specific medical instructions.
Skin: Immediately flood affected skin with water while removing and isolating all contaminated clothing. Gently wash all affected skin areas thoroughly with soap and water. Call a hospital or poison control center immediately, even if no symptoms develop.
Inhalation: Immediately leave the contaminated area and take deep breaths of fresh air. If symptoms develop (wheezing, coughing, shortness of breath, burning in the mouth, throat, or chest), call a physician and be prepared to transport the victim to a hospital. Rescuers entering an unknown atmosphere must use proper respiratory protection.
Ingestion: DO NOT INDUCE VOMITING. Corrosive chemicals can damage the membranes of the mouth, throat, and esophagus, and aspiration into the lungs during vomiting increases medical risks. If the victim is conscious and not convulsing, give 1 or 2 glasses of water to dilute the chemical and call a hospital or poison control center immediately. Transport the victim to a hospital immediately. If the victim is convulsing or unconscious, do not give anything by mouth, ensure the airway is open, and lay the victim on their side with the head lower than the body. DO NOT INDUCE VOMITING. Transport the victim IMMEDIATELY to a hospital.

Physical and Chemical Data
| Property | Value |
|---|---|
| Vapor Density | 4.88 (vs air) |
| Vapor Pressure | 1 mmHg (at 32 °C) |
| Autoignition Temperature | 1056 °F |
| Refractive Index | n20/D 1.553 (lit.) |
| Freezing Point | −2.0-0.0 °C |
| Density | 1.211 g/mL at 25 °C (lit.) |
| Functional Groups | Acyl chloride, phenyl |
| SMILES String | ClC(=O)c1ccccc1 |
| InChI | 1S/C7H5ClO/c8-7(9)6-4-2-1-3-5-6/h1-5H |
| InChI Key | PASDCCFISLVPSO-UHFFFAOYSA-N |
Applications
Benzoyl chloride is used in the synthesis of various organic building blocks, including:
- N,N-diethylbenzamide via condensation with diethylamine.
- N-2-bromophenylbenzamide by reacting with 2-bromoaniline via N-benzoylation.
- Propargyl benzoate via O-benzoylation of propargyl alcohol.
Benzoyl peroxide, used in the synthesis of polymers, is commercially synthesized by the reaction of benzoyl chloride, sodium hydroxide, and hydrogen peroxide.
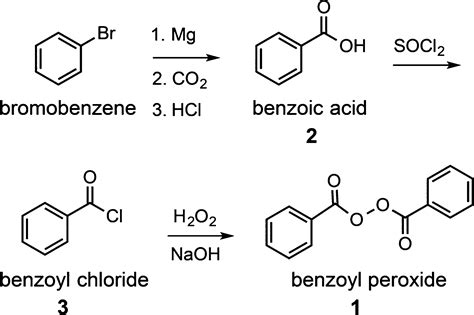
Hazard Classifications
According to GHS classifications, benzoyl chloride is:
- Acute Toxicity 3 (Inhalation)
- Acute Toxicity 4 (Dermal)
- Acute Toxicity 4 (Oral)
- Eye Damage 1
- Skin Corrosion 1B
- Skin Sensitization
- Reacts