COSENTYX® (secukinumab) offers various dosing options for patients aged 2 years and older with active psoriatic arthritis and for adults with moderate to severe plaque psoriasis. The choice between self-injection and intravenous (IV) infusion is a decision to be made with your doctor, considering what is best for your individual needs.
Administration Options: Self-Injection vs. IV Infusion
Your doctor can help you choose between self-injection at home or IV infusion in a medical office. This decision is based on your personal preference and medical suitability.
Self-Injection
Self-injection with COSENTYX provides a convenient once-a-month dosing schedule with no routine lab monitoring required during treatment for psoriatic arthritis (PsA). This can lead to fewer interruptions in your daily routine and offer relief from multiple PsA symptoms.
Starting COSENTYX Self-Injection
To begin treatment, your doctor will either prescribe 5 weekly doses or a single dose once a month, depending on your needs. After the initial loading doses, COSENTYX is administered once a month. The recommended dose is 150 mg, given as one subcutaneous injection.
Dosage Adjustments for Psoriatic Arthritis (PsA)
If you continue to experience PsA symptoms with the 150 mg dose, your rheumatologist may increase your dosage to 300 mg, administered as two 150 mg injections.
Dosage for PsA with Coexistent Plaque Psoriasis
For patients with PsA who also have moderate to severe plaque psoriasis, the recommended dosage is 300 mg, given as two 150 mg subcutaneous injections.
COSENTYX is administered using the Sensoready® Pen or a prefilled syringe. It is crucial to be checked for tuberculosis before starting treatment.
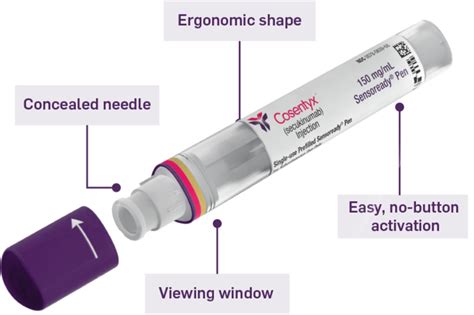
Using the Sensoready® Pen
The Sensoready® Pen is designed for safe and confident self-injection. It features:
- A concealed needle for safety.
- A viewing window to confirm the injection is complete.
- Easy, no-button activation with automatic 2-click technology to signal the start and end of the dose.
- An ergonomic shape for comfortable handling.
More than 90% of adults reported no pain or reaction during or after injection with the Sensoready Pen, and nearly 9 out of 10 adults expressed satisfaction with self-injection using this device. In clinical trials, very few adults (1.6%) experienced injection site reactions.
IV Infusion
If self-injection is not an option, your doctor may discuss COSENTYX IV infusion, which is a 30-minute infusion administered in a doctor's office or infusion center.
What to Expect with COSENTYX IV Infusion
COSENTYX is administered by a healthcare provider through an intravenous needle over approximately 30 minutes. The dosage is determined by your weight, and your healthcare provider will inform you about the frequency of infusions. If you miss an appointment, reschedule as soon as possible.
INTRATHECAL DRUG ADMINISTRATION AND CSF SAMPLE | ANIMATION |
Preparing for Your COSENTYX Infusion
Discuss with your healthcare provider what to expect, as infusion centers may vary. When preparing for your infusion:
- Hydrate well.
- Dress comfortably.
- Bring something to entertain yourself.
During the infusion, your healthcare provider may monitor your vital signs and answer any questions you have. After the infusion is complete, schedule your next appointment.
COSENTYX Dosing and Strengths by Indication
COSENTYX is available in different dosage forms and strengths for various indications:
Dosage Forms & Strengths
- Solution for SC injection: 150 mg/mL (single-dose Sensoready pen and prefilled syringe), 300 mg/2 mL (single-dose UnoReady pen and prefilled syringe).
- Solution for IV infusion: 125 mg/5 mL (single-dose vial; requires dilution, for adults only).
Adult Dosing
Plaque Psoriasis
- Indication: Moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
- Dosage: 300 mg subcutaneously (SC) at weeks 0, 1, 2, 3, and 4, followed by 300 mg SC every 4 weeks thereafter. For some patients, a dose of 150 mg SC may be acceptable.
Psoriatic Arthritis (PsA)
- Indication: Active psoriatic arthritis (PsA) in adults.
- Dosage (with coexistent moderate to severe plaque psoriasis): Use dosage and administration for plaque psoriasis (300 mg SC).
- Dosage (for other adults with PsA):
- With a loading dose: 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks.
- Without a loading dose: 150 mg SC every 4 weeks.
- IV Infusion (with loading dose): 6 mg/kg IV at week 0, then 1.75 mg/kg IV every 4 weeks (maintenance dose).
- IV Infusion (without loading dose): 1.75 mg/kg IV every 4 weeks. The maintenance dose should not exceed 300 mg per dose.
Ankylosing Spondylitis (AS)
- Indication: Active ankylosing spondylitis in adults.
- SC Administration (with loading dose): 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks. If active AS persists, consider increasing to 300 mg SC every 4 weeks.
- SC Administration (without loading dose): 150 mg SC every 4 weeks.
- IV Infusion (with loading dose): 6 mg/kg IV at week 0, then 1.75 mg/kg IV every 4 weeks (maintenance dose).
- IV Infusion (without loading dose): 1.75 mg/kg IV every 4 weeks. The maintenance dose should not exceed 300 mg per dose.
Non-radiographic Axial Spondyloarthritis (nr-axSpA)
- Indication: Active non-radiographic axial spondylarthritis with objective signs of inflammation.
- SC Administration (with loading dose): 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks thereafter.
- SC Administration (without loading dose): 150 mg SC every 4 weeks.
- IV Infusion (with loading dose): 6 mg/kg IV at week 0, then 1.75 mg/kg IV every 4 weeks (maintenance dose).
- IV Infusion (without loading dose): 1.75 mg/kg IV every 4 weeks. The maintenance dose should not exceed 300 mg per dose.
Hidradenitis Suppurativa (HS)
- Indication: Moderate to severe hidradenitis suppurativa (HS).
- Dosage: 300 mg SC at weeks 0, 1, 2, 3, and 4, followed by 300 mg SC every 4 weeks thereafter. If the response is inadequate, consider increasing the dose to 300 mg every 2 weeks. Each 300 mg dose can be given as one 300 mg injection or two 150 mg injections.
Pediatric Dosing (for patients aged 2 years and older)
Psoriatic Arthritis (PsA)
- Indication: Active psoriatic arthritis (PsA) in patients aged ≥2 years.
- Dosage (≥15 kg to <50 kg): 75 mg SC at weeks 0, 1, 2, 3, and 4, followed by 75 mg SC every 4 weeks.
- Dosage (≥50 kg): 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks.
Enthesitis-Related Arthritis (ERA)
- Indication: Active enthesitis-related arthritis (ERA) in patients aged ≥4 years.
- Dosage (≥15 kg to <50 kg): 75 mg SC at weeks 0, 1, 2, 3, and 4, followed by 75 mg SC every 4 weeks.
- Dosage (≥50 kg): 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks.
Plaque Psoriasis
- Indication: Moderate to severe plaque psoriasis in patients aged ≥6 years.
- Dosage (<50 kg): 75 mg SC at weeks 0, 1, 2, 3, and 4, followed by 75 mg SC every 4 weeks.
- Dosage (≥50 kg): 150 mg SC at weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks.
Hidradenitis Suppurativa (HS)
- Indication: Moderate to severe hidradenitis suppurativa (HS) in patients aged ≥12 years.
- Dosage (≥30 kg to <90 kg): 150 mg SC at Weeks 0, 1, 2, 3, and 4, followed by 150 mg SC every 4 weeks thereafter.
- Dosage (≥90 kg): 300 mg SC at Weeks 0, 1, 2, 3, and 4, followed by 300 mg SC every 4 weeks thereafter.
Important Dosing Considerations
Dosage Modifications
Studies have not been conducted to assess the effect of renal or hepatic impairment on COSENTYX pharmacokinetics.
Dosing Considerations
- Tuberculosis (TB) Evaluation: Patients should be evaluated for active or latent tuberculosis infection prior to initiating COSENTYX. COSENTYX initiation is not recommended in patients with active TB infection.
- Vaccinations: Complete all age-appropriate vaccinations as recommended by current immunization guidelines before initiating COSENTYX.
- Immune System Suppression: COSENTYX, like other biologics, suppresses the immune system and may increase the risk of infections. Tell your doctor if you have an active infection. Do not start treatment if you have a serious or recurrent infection (such as pneumonia).
- Inflammatory Bowel Disease (IBD): Caution should be used when prescribing secukinumab to patients with inflammatory bowel disease. Exacerbations, in some cases serious, occurred in secukinumab-treated patients during clinical trials.
Interactions and Contraindications
COSENTYX may interfere with the immune response to live vaccines. It is contraindicated with certain live vaccines due to the risk of infection. Before initiating COSENTYX, complete all age-appropriate immunizations. Non-live vaccinations received during treatment may not elicit an immune response sufficient to prevent disease.
Contraindicated Vaccines
COSENTYX is contraindicated with the following live vaccines:
- Adenovirus types 4 and 7 live, oral
- BCG vaccine live
- Influenza virus vaccine quadrivalent, intranasal
- Influenza virus vaccine trivalent, intranasal
- Measles, mumps, and rubella vaccine, live
- Measles, mumps, rubella, and varicella vaccine, live
- Rotavirus oral vaccine, live
- Smallpox and mpox (vaccinia) vaccine, live
- Typhoid polysaccharide vaccine
- Typhoid vaccine live
- Varicella virus vaccine live
- Yellow fever vaccine
- Zoster vaccine live
Serious Interactions with Vaccines
COSENTYX may decrease the effects of, or increase the risk of infection with, the following vaccines:
- Anthrax vaccine adsorbed
- Diphtheria & tetanus toxoids
- Diphtheria & tetanus toxoids/acellular pertussis vaccine
- Diphtheria & tetanus toxoids/acellular pertussis/poliovirus, inactivated vaccine
- Hepatitis A vaccine inactivated
- Hepatitis A/B vaccine
- Hepatitis B vaccine
- Human papillomavirus vaccine, nonavalent
- Influenza A (H5N1) vaccine
- Influenza A (H5N1) vaccine, adjuvanted
- Influenza virus vaccine quadrivalent
- Influenza virus vaccine quadrivalent, cell-cultured
- Influenza virus vaccine quadrivalent, recombinant
- Influenza virus vaccine trivalent
- Influenza virus vaccine trivalent, recombinant
It is recommended to avoid or use an alternate drug in conjunction with these vaccines, and to complete all age-appropriate immunizations prior to initiating COSENTYX.
Prescribing Approaches
Suggested SC Prescribing Approach for Appropriate Patients
For subcutaneous (SC) administration, it is recommended to separate loading dose and maintenance dose prescriptions. The last loading dose can be covered in the first maintenance-dose prescription.
Adult Patients
COSENTYX is intended for use under physician guidance. Adult patients can self-administer COSENTYX after proper training. The convenient SC dosing schedule allows for monthly maintenance doses.
Pediatric Patients
Pediatric patients should not self-administer COSENTYX. An adult caregiver should prepare and inject COSENTYX after receiving proper training.
Suggested Prescribing Approach for Plaque Psoriasis (PsO)
For adult patients with moderate to severe plaque psoriasis (PsO), with or without PsA:
- One prescription for loading doses (Weeks 0, 1, 2, and 3).
- One prescription for the maintenance dose, which covers the Week 4 loading dose as well as the ongoing maintenance dose.
For some patients, a loading and maintenance dose of 150 mg may be acceptable. Each 300 mg dose is given as 2 injections of 150 mg under the skin.
For pediatric patients aged 6 years and older with moderate to severe PsO:
- One prescription for loading doses (Weeks 0, 1, 2, and 3).
- One prescription for the maintenance dose, covering Week 4 and ongoing maintenance.
The recommended dosage for pediatric patients is based on body weight: 75 mg for those weighing <50 kg and 150 mg for those weighing ≥50 kg.
Preparation and Administration
Storage and Handling
COSENTYX must be kept refrigerated and should not be frozen. Allow the medication to warm to room temperature before injecting.
Injection Sites
Inject into the thigh, abdomen, or upper arm. Rotate injection sites with each administration.
Visual Inspection
Inspect COSENTYX visually for particulate matter and discoloration prior to administration. The solution should be clear to slightly opalescent, colorless to slightly yellow. Do not use if the liquid contains visible particles, is discolored, or cloudy.
IV Infusion Preparation
COSENTYX for intravenous use (solution in vials) requires dilution prior to administration. The diluted solution should be administered as soon as possible, and the total preparation and infusion time should not exceed 24 hours under refrigeration. Do not infuse COSENTYX concomitantly in the same intravenous line with other drugs.