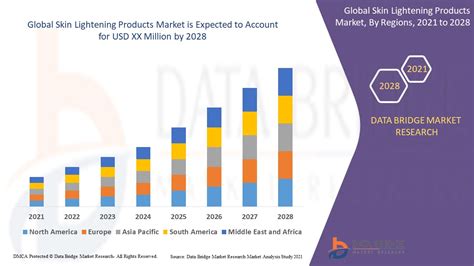
Skin lightening products, also referred to as skin brightening or fade products, are utilized by individuals of all skin tones to achieve a lighter complexion, diminish the appearance of freckles, or eliminate age spots. These products are widely available in various retail outlets, including stores catering to African, Asian, Middle Eastern, and Latino communities, as well as online marketplaces. The global market for skin lightening is substantial and projected to grow significantly, with spending expected to triple to US $31.2 billion by 2024, according to a report by Global Industry Analysts. In some regions, such as Nigeria, the usage of skin-lightening agents is particularly high, with 77% of women reportedly using them, followed by Togo (59%) and Senegal (27%).
Skin lightening and whitening agents in cosmetics are primarily used to create a more uniform skin tone, often targeting the face and neck, but sometimes applied to larger areas of the body. It is crucial to distinguish between a cosmetic product and a drug, as this distinction carries significant regulatory and safety implications. A product marketed as a cosmetic should not contain active pharmaceutical ingredients (APIs); otherwise, it falls under the classification of a drug according to regulations such as those set by the European Commission and the U.S. Food and Drug Administration (FDA).
Potential Health Hazards Associated with Skin Lightening Products
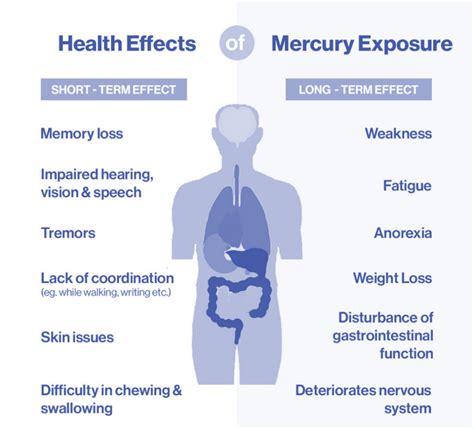
A significant concern regarding some skin lightening products is the presence of dangerous levels of mercury. Testing of these products in Minnesota revealed that certain formulations contained mercury levels far exceeding those permitted by the FDA for cosmetic products. While the FDA allows a maximum of one part per million (ppm) of mercury in cosmetics, the tested products ranged from 135 to 33,000 ppm. Exposure to mercury from these products can lead to serious health problems.
Individuals using skin products containing mercury have exhibited elevated mercury levels in their hair, blood, and urine. Mercury is known to cause damage to the kidneys, affect the nervous system, and induce skin pain or rashes. Particular caution is advised for pregnant individuals and those of child-bearing age, as well as for unborn babies, infants, and children, who are highly sensitive to mercury's toxic effects. Even in the absence of visible symptoms of mercury exposure in pregnant individuals, the mercury can still pose a risk to the unborn baby.
Identifying and Avoiding Mercury in Skin Lightening Products
To protect yourself from the dangers of mercury in skin lightening products, it is essential to carefully check product labels. If a product does not list its ingredients, it is advisable to discontinue its use. Furthermore, if the label explicitly states the presence of mercury, or uses related terms such as “calomel,” “mercuric,” “mercurous,” or “mercurio,” immediate cessation of use is recommended.
If possible, consumers should contact the product manufacturer directly to obtain a complete list of ingredients. If you have been using a product that you suspect contains mercury, stop using it immediately. The body naturally eliminates mercury over time. However, it is critical not to dispose of mercury-containing products in regular trash, as this can lead to environmental contamination and harm others. Such products should be taken to a household hazardous waste disposal site.
Mechanisms of Action and Regulatory Considerations
Skin lightening products, also known as bleaching creams, whiteners, skin brighteners, or fading creams, function by reducing the amount of melanin, a pigment responsible for skin coloration. Many individuals opt for these products to address specific skin concerns, including freckles, age spots, acne scars, or hormonal discoloration.
The analytical process for identifying and quantifying ingredients in cosmetic samples involves sophisticated techniques. For instance, samples can be analyzed using a Waters ACQUITY Arc UHPLC System equipped with a 2998 Photodiode Array (PDA) Detector and an ACQUITY QDa Mass Detector. The addition of mass detection complements PDA, enhancing the reliability of compound detection and identification. Through such analyses, several cosmetic samples have tested positive for prohibited skin lightening agents. Detailed methods for identifying skin lightening agents in cosmetics using UHPLC with PDA and mass detection, as well as rapid screening methods for corticosteroids like clobetasol propionate using direct analysis in real time (DART) with mass detection, are available.