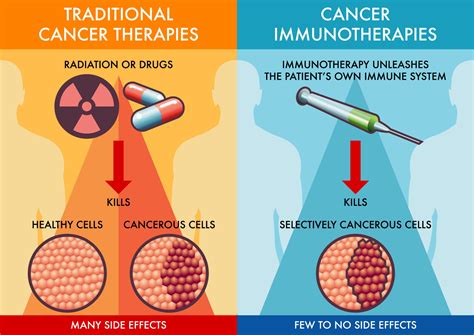
Immunotherapy is a revolutionary cancer treatment approach that harnesses the power of the body's own immune system to recognize and combat cancer cells. This innovative method has demonstrated significant efficacy, particularly in the treatment of advanced melanoma.
Understanding Immunotherapy
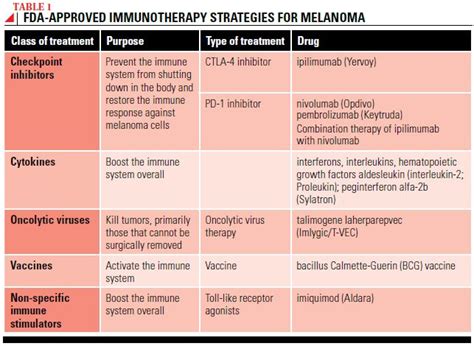
At its core, immunotherapy utilizes drugs to empower the immune system, enabling it to identify and fight cancerous growths. Among the various forms of immunotherapy, checkpoint inhibitors are the most commonly employed for melanoma treatment. These drugs function by releasing the natural "brakes" on T cells, allowing these crucial immune cells to seek out and destroy tumors. This mechanism is also referred to as immune checkpoint blockade, as the drug intervenes in the molecular checkpoints that normally regulate T cell activity.
Currently, three primary checkpoint inhibitor drugs are available for advanced melanoma:
- Ipilimumab (Yervoy®)
- Nivolumab (Opdivo®)
- Pembrolizumab (Keytruda®)
Ipilimumab (Yervoy®) for Advanced Melanoma
Ipilimumab is a potent immunotherapy option for individuals with metastatic melanoma and stage III melanoma that cannot be completely removed through surgery. Its mechanism of action involves blocking an immune molecule known as CTLA-4. Notably, patients at Memorial Sloan Kettering (MSK) were among the first globally to receive ipilimumab treatment in 2004. MSK played a pivotal role in early clinical studies that established ipilimumab's ability to prolong overall survival in metastatic melanoma patients. The U.S. Food and Drug Administration (FDA) approved ipilimumab for general use in 2011, with MSK patients having access to it through clinical trials years prior.
Anti-PD-1 Therapy for Advanced Melanoma
Nivolumab (Opdivo®) and pembrolizumab (Keytruda®) are classified as PD-1 blockers. These drugs work by inhibiting the PD-1 molecule, thereby enhancing the immune system's ability to fight advanced and stage III melanoma, especially when surgical removal is incomplete. Even in cases where stage III melanoma is surgically resectable, anti-PD-1 immunotherapy may offer significant benefits. Early clinical research for these drugs was largely conducted at MSK, with patients having access through clinical trials since 2010, approximately four years before FDA approval.
While research into anti-PD-1 therapies is ongoing, they have shown considerable success. Many patients experiencing significant benefits can stop treatment with a low rate of melanoma recurrence. Furthermore, individuals whose disease progresses after discontinuing anti-PD-1 therapy may still respond if treatment is reinstituted.
Cancer Immune Checkpoint Inhibitors
Combination Immunotherapy for Advanced Melanoma
An emerging strategy for advanced melanoma involves the combination of ipilimumab and nivolumab. This approach is based on the premise that these two drug classes are more effective when used together than individually. MSK was the first institution to investigate this combination in patients, leading pivotal clinical trials that resulted in the FDA's approval of this combination therapy in 2015.
Understanding Stage III Melanoma
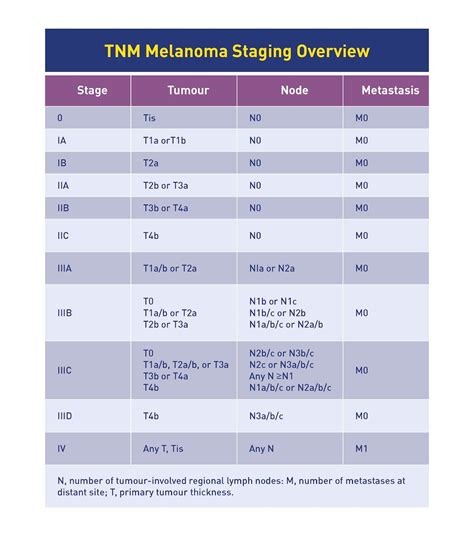
Stage III melanoma is characterized by the spread of the tumor to regional lymph nodes or the development of in-transit deposits, without evidence of distant metastasis. This stage signifies regional spread, meaning the cancer has moved from the primary site to nearby lymph nodes but not to distant organs. Stage III melanoma is further categorized into four subgroups: IIIA, IIIB, IIIC, and IIID, defined by the TNM (Tumor, Node, Metastasis) staging system.
TNM Staging Explained
- T (Tumor): Describes the primary tumor's thickness.
- T1: < 1.0 mm thick
- T2: 1.0 mm to 2.0 mm thick
- T3: 2.0 mm to 4.0 mm thick
- T4: > 4.0 mm thick
- N (Node): Relates to the spread to regional lymph nodes.
- N1: Spread to one lymph node, or in-transit/satellite/microsatellite metastasis.
- N2: Spread to two or three lymph nodes, or a combination of lymph node spread and in-transit/satellite/microsatellite metastasis.
- N3: Spread to four or more lymph nodes, or extensive lymph node involvement with matted nodes.
- M (Metastasis): Indicates the presence of distant metastasis (absent in Stage III).
Subgroups within Stage III are determined by specific combinations of T and N categories. For instance, T2aN2a indicates a tumor greater than 1.0 mm but up to 2.0 mm thick that was not ulcerated, with spread to two to three clinically occult lymph nodes.
Treatment Options for Stage III Melanoma
Despite surgical treatment, Stage III melanoma carries an intermediate to high risk of local recurrence or distant metastasis. Treatment options are diverse and may include surgery (such as sentinel lymph node biopsy and potentially completion lymph node dissection), neoadjuvant therapy, adjuvant therapy, radiation therapy, and participation in clinical trials. Patients typically consult with both surgical and medical oncologists.
Surgical Interventions
- Wide Local Excision: The standard primary treatment for melanoma, involving the removal of the primary tumor with surrounding tissue.
- Sentinel Lymph Node Biopsy (SLNB): Performed to determine if cancer has spread to the nearest lymph nodes.
- Completion Lymph Node Dissection (CLND): May be recommended if cancer is found in the sentinel lymph node, involving the removal of remaining lymph nodes in the area. However, recent studies suggest CLND may not always prolong survival.
- Therapeutic Lymph Node Dissection (TLND): Surgery to remove all lymph nodes in an area when lymph nodes are enlarged or detected on examination or imaging.
Systemic Therapies
- Neoadjuvant Therapy: Administered before primary treatment (usually surgery) to shrink tumors. For Stage III patients, this is often part of clinical trials.
- Adjuvant Therapy: Given after primary treatment to eliminate any remaining cancer cells and reduce the risk of recurrence. This is often a systemic treatment.
Specific Immunotherapy Drugs for Melanoma
Pembrolizumab (Keytruda®)
Purpose: Keytruda is a humanized monoclonal antibody designed to block the PD-1 cellular target, thereby unleashing the immune system against melanoma cells.
How it works: By inhibiting the interaction between PD-1 and its ligands (PD-L1 and PD-L2), Keytruda enhances the body's anti-tumor immune response.
Indications: Approved for Stage IIB, IIC, III, and IV melanoma patients, including those following complete resection of Stage IIB/IIC and Stage III melanoma.
Administration: Given intravenously, typically every 3 weeks, until disease progression or unacceptable side effects.
Effectiveness: Clinical trials have shown tumor shrinkage in a significant percentage of patients, with effects lasting for extended periods. Ongoing studies continue to evaluate its efficacy.
Side Effects: Keytruda can trigger immune system reactions affecting various organs, which can be serious or life-threatening. Common side effects include fatigue, cough, nausea, itching, rash, decreased appetite, constipation, and joint pain. Severe side effects can involve lung, intestinal, liver, kidney, and hormone gland problems.

Nivolumab (Opdivo®)
Purpose: Opdivo is a humanized monoclonal antibody that targets PD-1, leading to an enhanced anti-tumor immune response.
How it works: By blocking PD-1, Opdivo releases the inhibition of the immune response, including the anti-tumor response.
Indications: Approved for Stage III patients with lymph node involvement after complete resection, and for Stage IV melanoma.
Administration: Given intravenously every 2 or 4 weeks for up to 1 year in Stage III patients, or until disease progression in Stage IV patients.
Effectiveness: Clinical trials have demonstrated a significant reduction in the risk of recurrence or death for Stage III patients and a reduction in disease progression for Stage IV patients compared to ipilimumab. Long-term survival rates have also been reported.
Side Effects: Similar to Keytruda, Opdivo can cause immune-mediated side effects affecting various organs. These can include lung, intestinal, liver, kidney, and hormone gland problems. Common side effects include fatigue, cough, chest pain, diarrhea, nausea, and itching.
Ipilimumab (Yervoy®)
Purpose: Yervoy is a human monoclonal antibody designed to block CTLA-4, a protein that regulates T cell activation. It restores and strengthens the immune system's ability to fight cancer.
How it works: By blocking CTLA-4, Yervoy "releases the brakes" on the immune system, allowing T cells to activate and attack melanoma cells.
Indications: Approved for Stage III or IV melanoma patients. For adjuvant treatment, it is given after complete surgical removal of melanoma to lower the risk of return.
Administration: Given intravenously. For adjuvant melanoma, a specific dosage regimen is followed for up to 3 years. For unresectable or metastatic melanoma, a different dosage regimen is administered for a total of 4 doses.
Effectiveness: Clinical trials have shown significant improvements in overall survival for patients treated with Yervoy, both in combination with other treatments and as a standalone therapy. Long-term survival rates have also been observed.
Side Effects: As with other immunotherapies, Yervoy can cause immune-related side effects affecting various organs. These can include gastrointestinal issues, skin reactions, and inflammation of endocrine glands.
Combination Therapy: Ipilimumab and Nivolumab
The combination of ipilimumab and nivolumab is a powerful approach for advanced melanoma, leveraging the distinct mechanisms of both drugs. This combination has shown greater efficacy than either drug used alone. MSK led the clinical trials that supported the FDA approval of this combination therapy in 2015.
Side Effects of Immunotherapy
Side effects are a critical consideration in immunotherapy decisions. Combination therapies, such as ipilimumab plus nivolumab, may present more severe side effects than monotherapy. Experts in immunotherapy are adept at identifying patients at higher risk for side effects and managing them effectively. Ongoing research aims to develop new strategies for preventing and managing these side effects.
Patients should discuss the potential benefits and risks of any immunotherapy treatment with their doctor. Common side effects across these therapies can include fatigue, rash, diarrhea, nausea, itching, and flu-like symptoms. More severe side effects involve inflammation in organs like the lungs, intestines, liver, kidneys, and endocrine glands. Prompt medical attention is crucial for managing these potential issues.
Cancer Immune Checkpoint Inhibitors
Clinical Trials and Future Directions
Memorial Sloan Kettering (MSK) is actively involved in studying how immunotherapy can benefit more melanoma patients. This includes clinical trials testing novel combinations of immunotherapy treatments and exploring new drugs for patients whose melanoma has not responded to anti-PD-1 therapy. MSK patients may have the opportunity to enroll in these trials, offering access to promising new approaches.
The field of immunotherapy for melanoma is continuously evolving. While it has become a standard of care treatment in the last decade, research is ongoing to optimize treatment delivery, understand factors influencing patient response, and identify new therapeutic targets. Studies are exploring immunotherapy optimization trials to determine ideal dosing and schedules, potentially reducing side effects and improving long-term outcomes. Furthermore, research into the gut microbiome's influence on immunotherapy response and the potential role of circulating tumor DNA (ctDNA) in guiding adjuvant treatment decisions highlights the dynamic nature of this field.
The ultimate goal of immunotherapy care is not only to cure or control the cancer but also to ensure the best possible quality of life for each patient.
tags: #immunotherapy #for #stage #3 #melanoma