Melanoma, the most prevalent form of skin cancer, presents a significant challenge, particularly for patients with advanced stages who no longer respond to standard immunotherapies. Treatment options for these individuals are severely limited. However, recent research from Vanderbilt, led by Professor Emerita of Pharmacology Ann Richmond, has unveiled a promising new drug combination. This innovative approach targets three crucial pathways simultaneously, potentially re-enabling these treatment-resistant patients to respond to their body's own immune defenses.
Melanoma affects over 300,000 people globally, with an increasing incidence in recent years. Metastatic melanoma tumors, which have spread from their original site, frequently develop resistance to immune checkpoint inhibitors (ICIs). ICIs are a type of immunotherapy designed to remove the molecular "brakes" that prevent immune cells from attacking tumors, and they represent the standard of care for metastatic melanoma. For patients with specific mutations, such as those in the BRAF gene, targeted therapies like BRAF/MEK inhibitors are available. These drugs block the MAPK signaling pathway, which drives tumor growth. While beneficial for some, treatment options are particularly scarce for patients without these specific mutations.
Another avenue of research involves exploring novel drug combinations. A class of drugs known as CDK inhibitors has shown potential in slowing tumor growth and boosting the immune system, especially when used in conjunction with other treatments. Researchers are actively investigating innovative combinations to address the limitations of current therapies. Furthermore, a promising strategy involves targeting immune-suppressing cells within the tumor environment using CXCR1/2 blockers, which can aid the body's T-cells in combating cancer.
Innovative Drug Combinations and Strategies
Professor Emerita Ann Richmond highlighted a significant advancement: "This is the first study combining both CDK4/6 and CDK2 inhibitors with a chemokine receptor antagonist to treat melanoma tumors." Chemokine receptor antagonists are drugs that block specific proteins acting as "signals" to attract immune cells to diseased areas. The research demonstrated that blocking CDK4/6 and CDK2 enhances the recruitment of T-cells into the tumor. However, it was also observed that myeloid-derived suppressor cells (MDSCs), which suppress the body's anti-tumor immune response, can inhibit T-cell activity.
The translational promise of the tested drug combination is considered high, with a potential for accelerated progression to human studies, as all three drugs are already undergoing clinical trials for other conditions. This groundbreaking study was a collaborative effort involving Vanderbilt and international partners, with significant contributions from Vanderbilt scientists Jinming Yang, Weifeng Luo, Patricia Ward, and Chi Yan. Yang and Luo are recognized as equal first authors for their contributions.
The Evolving Landscape of Melanoma Immunotherapy
The field of melanoma treatment is rapidly expanding its arsenal of immune therapies. Several are currently approved, while others are undergoing testing for patients with advanced disease. Some are also being utilized in adjuvant (post-surgical) or neoadjuvant (pre-surgical) settings. Approved immune therapies can be broadly categorized based on their mechanism of immune system activation and their ability to enable immune cells to identify and destroy melanoma cells. These categories include:
- Cytokine therapy: such as interleukin 2 (IL-2) and aldesleukin.
- Immune checkpoint inhibitors: including anti-PD-1, anti-CTLA4, anti-LAG3 antibodies, and their combinations.
- Oncolytic viral therapies: such as T-VEC.
- Bispecific proteins: which bridge tumor and immune cells, like Tebentafusp.
- Cell-based therapy: commonly known as tumor-infiltrating lymphocytes (TILs).
Many of the immune therapies pioneered in melanoma are now being applied or tested in over 30 other types of cancer. Despite these remarkable advancements, approximately 50% of patients do not benefit from these novel treatments, creating an urgent need for improvements in existing immunotherapeutic approaches or the discovery of new methods to activate the immune system.
Clinical Trials and Promising New Approaches
In recent years, encouraging early clinical trial data has facilitated the advancement of several investigational immune therapies into Phase 2 and Phase 3 clinical trials for further evaluation. These trials aim to gather comprehensive data on treatment effectiveness, safety, and side effects in larger patient populations. The "Melanoma Clinical Trials to Watch" series highlights three distinct trials currently enrolling patients, focusing on a new immune checkpoint combination and two different cell-based therapy approaches.
New Checkpoint Combination: Phase 3 Harmony Head to Head Trial
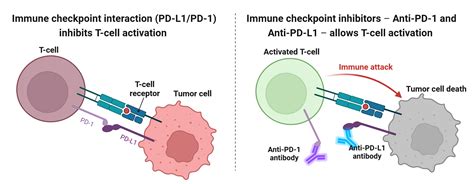
Immune cells express checkpoint molecules that act as brakes on the immune system's ability to detect and eliminate cancer cells. Immune checkpoint inhibitors (ICIs) are antibody treatments that bind to and block these checkpoints, thereby releasing the brakes and allowing immune cells to destroy cancer cells. Different ICI therapies have been developed for three well-characterized immune checkpoints: PD-1, CTLA-4, and LAG-3.
Approved ICIs such as pembrolizumab (Keytruda) and nivolumab (Opdivo) block the PD-1 immune checkpoint molecule. Both are approved for advanced and metastatic melanoma, as well as for adjuvant treatment of resectable melanoma. Combinations of anti-PD-1 drugs with other immune checkpoint drugs are being tested to assess their potential for enhanced anti-tumor activity compared to anti-PD-1 monotherapy. Based on Phase 2/3 clinical trial results, the combination of nivolumab with relatlimab (blocking the LAG-3 checkpoint) received FDA approval for advanced melanoma in 2022.
The biotechnology company Regeneron is evaluating a combination of a novel LAG-3-blocking drug, fianlimab, and a PD-1-blocking drug, cemiplimab (Libtayo®). A Phase 1 clinical trial (NCT03005782) involving 98 patients with advanced melanoma showed promising results: 57% of patients responded (indicating tumor shrinkage), with 25% achieving a complete response (cancer disappearance). The safety profile of the fianlimab and cemiplimab combination was comparable to single-agent anti-PD-1 therapies.
Building on these encouraging Phase 1 data, the Phase 3 Harmony Head to Head trial (NCT06246916) is designed to compare the safety and efficacy of the combination of fianlimab and cemiplimab against the approved combination of relatlimab and nivolumab. This trial is currently enrolling patients aged 18 and older with unresectable (Stage 3) or metastatic (Stage 4) melanoma who have not received prior systemic treatments. Patients must provide informed consent and undergo screening to determine eligibility. Eligible patients will be randomly assigned to receive either the combination of fianlimab and cemiplimab every 3 weeks or the combination of relatilimab and nivolumab every 4 weeks. Treatment duration will depend on drug efficacy and patient tolerance, with regular medical assessments.
New Cell-Based Therapy Approaches
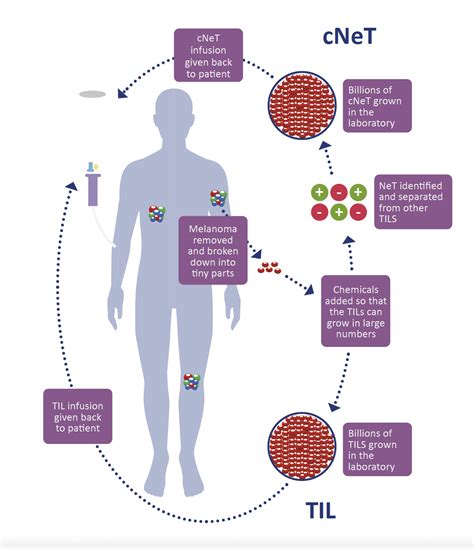
Cellular therapies utilize living cells to enhance the immune system's response and fight cancer. In 2024, AMTAGVI (lifileucel) became the first cellular therapy approved for a solid cancer, specifically melanoma. AMTAGVI is a tumor-infiltrating lymphocyte (TIL) therapy that uses T-lymphocyte cells isolated from surgically removed tumor tissue. These cells are then multiplied extensively in a laboratory and activated before being infused back into the patient. Prior to infusion, patients undergo a conditioning regimen of chemotherapy (lymphodepletion) to suppress their immune system, creating space for the infused T-cells to expand and function effectively. Following infusion, a specific dosage of the cytokine IL-2 (aldesleukin) is administered to activate the infused T-cells.
Next Generation TILs: Agni-01 Trial
Newer versions of TILs are under development, involving genetic modifications of the isolated T-cells before infusion. The company Obsidian is engineering a TIL therapy (OBX-115 TIL) that is equipped with a protein called IL15, expressed on the surface of the engineered TILs (membrane-bound IL15 or mbIL15). mbIL15 has the potential to enhance the anti-tumor activity of modified TILs and lead to more durable responses. Furthermore, the expression of mbIL15 in TILs can be regulated by acetazolamide (ACZ), an FDA-approved drug. ACZ allows for the activation of infused T-cells and can be administered to boost TIL function when needed, or reduced if side effects occur. This approach also eliminates the need for IL-2 infusions, which some patients cannot tolerate.
Promising data from the Agni-01 Phase 1/2 trial (NCT06060613) were presented at the 2025 American Society of Clinical Oncology annual conference. This trial evaluated the regimen of OBX-115 TIL in patients who had not responded to immune checkpoint inhibitor therapy (ICI-resistant). Phase 1 trials focus on dose escalation to determine the optimal drug dose for Phase 2 trials, which then concentrate on efficacy and further safety evaluation.
The Agni-01 trial is enrolling patients aged 18 and older with melanoma that is unresponsive to or has progressed following ICI treatment. The treatment protocol includes tumor procurement by surgery or biopsy to isolate TILs, low-dose lymphodepletion to suppress the immune system, infusion of modified TILs (engineered with mbIL15), and administration of ACZ. The trial is accessible at multiple sites.
TCR-T Cell Based Therapy - SUPREME Phase 3 Trial
TCR-T cell-based therapy is a form of cancer immunotherapy where T-cells, isolated from a patient's blood, are genetically engineered to express a T-cell receptor (TCR) that recognizes cancer-specific protein fragments (peptide antigens). These peptide antigens are bound to a specific marker, HLA-A*02:01, and presented on the surface of tumor cells. The engineered TCR recognizes this peptide antigen/marker complex, prompting the T-cells to target and eliminate cancer cells.
The biotechnology company Immatics is developing a TCR-T therapy (IMA203) that targets the protein PRAME, which is highly expressed in various solid tumors, including melanoma. The engineered PRAME TCR-T recognizes PRAME-specific peptide antigens linked to the HLA-A*02:01 marker displayed on tumor cells, initiating tumor cell destruction. Results from a Phase 1b trial involving 33 patients with metastatic melanoma who had failed prior therapies showed favorable tolerability and promising clinical activity with a single infusion of IMA203 PRAME cell therapy. The objective response rate (cORR), defined as sustained shrinkage or disappearance of melanoma, was 56%. Among these patients, nearly half had cutaneous melanoma and the other half had uveal melanoma (melanoma of the eye), with cORR rates of 50% and 67%, respectively, for each subtype.
The SUPREME Phase 3 trial (NCT05577267) is being conducted in collaboration with Immatics and is enrolling patients aged 18 and older with unresectable or metastatic cutaneous melanoma who have progressed on prior therapies. Participants must also test positive for the HLA-A*02:01 marker. Patients with acral melanoma may also be eligible if they meet the criteria. In this trial, eligible patients will be randomly assigned to either Group 1 (receiving the IMA203 regimen) or Group 2 (receiving investigator's choice of selected approved therapies).
Advances in Immunotherapy and Targeted Treatments
Immunotherapy broadly refers to treatments that stimulate a patient's immune system to attack cancer cells. Immune checkpoint blockers interfere with the signals cancer cells use to evade immune detection. By interrupting these inhibitory signals, the immune system can be unleashed to attack cancer.
For some cancers, a single checkpoint blocker may be sufficient to enable immune cells to attack. In other cases, a combination of two or three checkpoint blockers might be necessary. Recent research has also explored the adjuvant (post-surgical) use of anti-PD-1 drugs or BRAF/MEK inhibitors (for BRAF-mutant melanomas), demonstrating an extended disease-free period for patients with Stage III or limited Stage IV melanomas. These findings have led to several FDA approvals, establishing adjuvant treatments for melanoma as standard care.
Researchers are also investigating the neoadjuvant (pre-surgical) application of anti-PD-1 therapies for earlier stages of skin cancer. The close collaboration between laboratory researchers and clinicians is identified as a key driver of advances in immunotherapy, aiming to tailor treatments for long-lasting cancer control.
Skin cancers generally exhibit a high number of DNA mutations, often attributed to ultraviolet light exposure. Many of these cancers also express the PD-L1 protein. However, some skin cancers with high mutation burdens and PD-L1 expression do not respond to immunotherapy. Understanding these biomarkers and uncovering new ones is a major focus of ongoing research.
Adoptive cell therapy represents another potent immunotherapy for melanoma. In this approach, immune cells within the patient's tumor, known as tumor-infiltrating lymphocytes (TILs), are isolated, modified in a laboratory to enhance their cancer-fighting capabilities, and then re-infused into the patient. Melanomas with specific BRAF gene mutations may respond to BRAF and MEK inhibitors.
A recent Phase 3 randomized trial indicated that immunotherapy should be recommended as the initial treatment for most patients diagnosed with advanced melanoma, given the potential for long-lasting remissions. While basal cell carcinoma is the most common skin cancer and typically curable with simple procedures, a small subset of patients develop advanced or metastatic disease. Standard treatments for these cases can have significant side effects and limited long-term efficacy. Researchers are now studying anti-PD-1 (nivolumab, Opdivo) for advanced basal cell skin cancer as a first-line treatment or for recurrent disease.
Experts have also co-led national trials demonstrating the efficacy of anti-PD-1 (pembrolizumab, Keytruda) in advanced Merkel cell carcinoma (MCC), a rare and aggressive skin cancer. This led to the FDA approval of pembrolizumab for advanced MCC in 2018, establishing a new standard of care. Further research showed that neoadjuvant anti-PD-1 (nivolumab, Opdivo) could induce complete tumor regression in approximately 50% of patients with MCC, leading to long-lasting results. This combination of immunotherapy with surgery has influenced clinical practice.
In kidney transplant recipients, skin cancer is the most common cancer, often developing years after transplantation due to the immune suppression required to prevent organ rejection. Current research is exploring an innovative clinical trial of immunotherapy for kidney transplant recipients with advanced skin cancers, combining anti-PD-1 immunotherapy (nivolumab) with immunosuppressant drugs used for kidney rejection prevention. This multicenter collaboration is supported by the National Institutes of Health.
The Johns Hopkins Melanoma Program offers comprehensive expertise in managing melanoma at all stages. Specialists collaborate with researchers and national cooperative groups to develop and conduct studies of experimental therapies. Interferon is an FDA-approved therapy for surgically removed melanoma tumors. Interleukin 2 (IL-2) activates immune cells to fight cancer and is administered as a five-day inpatient treatment, potentially repeated based on tumor response. Johns Hopkins is one of the few Comprehensive Cancer Centers equipped to administer IL-2 therapy.
Ipilimumab (Yervoy) is administered intravenously every three weeks for a total of four doses and has shown to shrink tumors and improve survival in some melanoma patients. Investigators at Johns Hopkins are also developing novel drugs to restore the immune system's ability to detect and attack cancer cells.
Targeting Resistance and Improving Treatment Outcomes
Some melanomas harbor genetic mutations that drive tumor growth. A new study offers potential breakthroughs for patients with advanced melanoma who do not respond to current immunotherapies. Immunotherapy activates the body's immune system against cancer, but a significant percentage of patients either do not respond or relapse over time.
Researchers have tested combinations of drugs that block immune checkpoints, such as PD-1, LAG-3, and TIM-3, which regulate immune responses. A team led by Keiran Smalley, Ph.D., director of Moffitt's Donald A. Adam Melanoma and Skin Cancer Centre of Excellence, found that a triple checkpoint combination was particularly effective in models that had not responded to standard therapies. TIM-3 is often found on immune cells that are too exhausted to fight cancer effectively, and analysis of melanoma patient tumor samples revealed higher TIM-3 expression in those who did not respond to immunotherapy. These findings suggest a new approach for treating melanoma patients with limited options.
Unresectable or metastatic melanoma is an aggressive and potentially fatal cancer. While melanomas constitute only about 1% of all skin cancers, they account for a substantial number of cancer-related deaths. Treatments for advanced or metastatic melanoma may include immunotherapy using PD-1 inhibitors, which help the immune system fight cancer cells, and drugs targeting the BRAF gene for melanomas with specific mutations.
Amtagvi is a tumor-derived autologous T cell immunotherapy. It involves surgically removing a portion of the patient's tumor tissue, isolating and manufacturing the T-cells, and then re-infusing them into the patient as a single dose. Amtagvi was approved via the FDA's Accelerated Approval pathway, which allows for earlier access to promising therapies for serious illnesses with unmet medical needs, based on surrogate endpoints that predict clinical benefit.
The safety and effectiveness of Amtagvi were evaluated in a global, multicenter clinical study of adult patients with unresectable or metastatic melanoma who had previously received at least one systemic therapy, including a PD-1 blocking antibody and, if applicable, a BRAF inhibitor. The objective response rate was 31.5%, with 4.1% achieving a complete response and 27.4% a partial response. Potential risks associated with Amtagvi include prolonged severe low blood counts, severe infections, cardiac disorders, worsened respiratory or renal function, and fatal treatment-related complications. A Boxed Warning in the drug's label highlights these risks, and patients require close monitoring.
Immunotherapy is often the first-line treatment for advanced melanoma. However, its effectiveness may diminish when the tumor spreads to the brain, or as patients develop resistance over time. Research is exploring the inhibition of the enzyme FAK in combination with RAF and MEK inhibitors, which target another cellular pathway regulating cancer cell growth. This combination therapy has shown effectiveness in prolonging survival rates in preclinical models and has demonstrated an ability to halt the development of brain metastases.
While melanoma is relatively rare, its incidence has been rising rapidly, and it accounts for the majority of skin cancer deaths. However, advancements in immunotherapy have dramatically improved survival rates for many patients. Pioneering research has transformed melanoma from a largely untreatable disease to one with a significant cure rate for responding patients. Despite these successes, immunotherapy still does not work for everyone, highlighting the need to identify mechanisms of resistance and predict patient response.
To enhance therapy response rates, researchers are developing new combination therapies. Combining checkpoint inhibitors with other agents has shown greater benefits for some patients. For instance, combining two immune checkpoint inhibitors-ipilimumab and nivolumab-has been shown to overcome resistance to prior PD-1 therapy in patients with metastatic melanoma, suggesting this combination as a preferred regimen for those unresponsive to prior immunotherapy.
Another study explored a triple combination therapy using BRAF and MEK inhibitors along with an immune checkpoint inhibitor for advanced melanoma. Results indicated that patients receiving this combination as initial treatment survived longer without cancer progression. An early-stage study also found an effective combination of the immunotherapy drug pembrolizumab with an experimental agent that mimics a bacterial infection. This therapy altered the tumor microenvironment, enabling a more effective immune attack. Early results showed tumor shrinkage in a subset of patients.
CAR T-cell Therapies and Future Directions
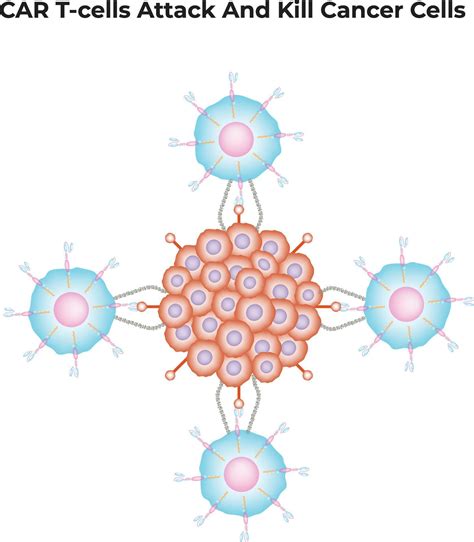
Building on these developments, researchers are exploring the potential of chimeric antigen receptor (CAR) and T-cell receptor (TCR) gene-engineered T-cell therapy for rare and difficult-to-treat melanomas. CAR T-cell therapy involves engineering a patient's immune cells to express synthetic receptors that enable them to identify and attack tumor cells based on specific surface proteins. One area of investigation involves using CAR T-cell therapy to treat patients whose tumors express the protein IL13Ra2, which is commonly found in melanoma and other cancers.
Another approach involves using CAR T-cells to target rare forms of melanoma, such as acral, uveal, and mucosal melanoma. This new strategy is designed to recognize and attack cells with high levels of TYRP1, a protein found on the surface of melanoma cells. Preclinical tests have shown that these engineered CAR T-cells can effectively eliminate cancer cells without causing severe side effects. These advancements are part of a broader vision to offer more targeted and effective treatments for patients with melanoma and other challenging cancers.
Tumor-infiltrating lymphocyte (TIL) therapy has shown remarkable success in patients with advanced melanoma. One patient, Pattie Fuller, whose melanoma had progressed to stage 4 and spread to multiple organs, experienced complete disappearance of tumors in her lungs and liver and significant shrinkage in lymph nodes after receiving TIL therapy (lifileucel, Amtagvi®) at Memorial Sloan Kettering Cancer Center (MSK). TIL therapy involves isolating immune cells from a patient's tumor, multiplying them in a lab, and re-infusing them after a conditioning chemotherapy regimen.
MSK conducted clinical trials that provided evidence for the FDA approval of lifileucel, and has since treated numerous patients with this therapy. The process for Pattie involved minimally invasive surgery to collect TIL cells, followed by a period at home while the cells were grown. She then returned for treatment, experiencing minimal side effects. MSK provided comprehensive support, including accommodation and a caregiver, during her recovery period.
During her treatment, Pattie discovered she had inherited a gene for Lynch syndrome, a hereditary disorder increasing the risk of several cancers, including melanoma, colorectal, and uterine cancer. She is now undergoing regular screenings for these associated cancers and has informed her family members about their potential risk. Pattie actively shares her melanoma survival story and her positive experience with the care received at MSK.