Calcium is a vital mineral in the human body, playing a critical role in numerous physiological functions. Maintaining optimal calcium levels is crucial for bone health, muscle contraction, nerve transmission, and blood clotting. Abnormalities in calcium levels, whether too high (hypercalcemia) or too low (hypocalcemia), can lead to various health issues and necessitate further evaluation and management by healthcare providers. Regular monitoring of calcium levels through routine blood tests helps assess overall health and identify potential imbalances early on, enabling timely intervention and appropriate management to maintain calcium homeostasis.
When blood calcium levels are assessed, it's important to understand that calcium exists in different forms in the bloodstream. Roughly half of the calcium is bound to proteins, primarily albumin, while the other half is free, or ionized. The total blood calcium level measures both bound and free calcium. However, in certain conditions, such as malnutrition or kidney disease, where blood protein levels may be low, the total blood calcium level might appear falsely low. In such cases, measuring ionized calcium, also known as free calcium, becomes more accurate as it reflects the metabolically active form of calcium available for use by the body.
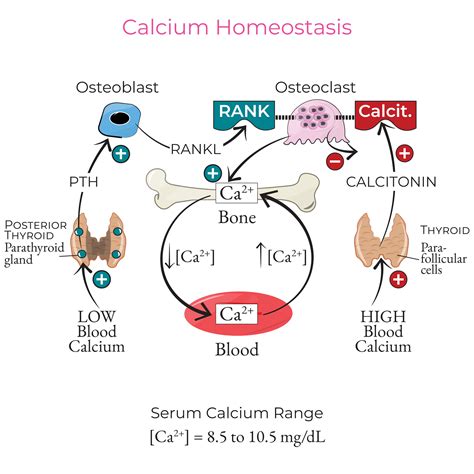
Understanding Total and Ionized Calcium
Total serum calcium is the most commonly measured parameter. It includes calcium bound to proteins, primarily albumin, and calcium complexed with anions like bicarbonate and phosphate, as well as the free, ionized fraction. The normal range for total blood calcium is typically between 8.5 to 10.4 mg/dL, though this can vary slightly between laboratories. Factors such as medications (e.g., diuretics) and certain health conditions (e.g., kidney disease, malnutrition) can influence these normal ranges.
Ionized calcium (iCa2+), on the other hand, represents the metabolically active form of calcium that is not bound to proteins. This free calcium is what the body's cells and regulatory systems, like the parathyroid glands, directly sense and respond to. Its concentration is critical for cellular functions, including nerve impulse transmission and muscle contraction.
The Role of Albumin in Calcium Binding
Albumin is the primary protein responsible for binding calcium in the blood. Under normal physiological conditions, about 35-45% of total serum calcium is protein-bound, with approximately 90% of this bound to albumin. Each albumin molecule has multiple binding sites for calcium. This protein binding acts as a buffer, moderating the immediate impact of acute calcium loads on ionized calcium levels.
However, changes in serum albumin concentration can significantly affect the total serum calcium measurement. In conditions of hypoalbuminemia (low albumin levels), the total serum calcium may appear falsely low because less calcium is bound to proteins. The total calcium level tends to decrease by about 0.8 mg/dL for every 1 g/dL reduction in serum albumin. For this reason, it is crucial to measure serum albumin alongside serum calcium for accurate interpretation, especially in patients with suspected malnutrition or kidney disease.
Challenges with Albumin-Adjusted Calcium Formulas
Historically, formulas like the Payne formula (Albumin-adjusted calcium (mmol/L) = Total calcium (mmol/L) + 0.02 [40 - albumin (g/L)]) have been used to adjust total calcium measurements for low albumin levels. The rationale behind these formulas is to estimate what the calcium level would be if the albumin concentration were normal, thereby providing a more accurate reflection of ionized calcium. However, the clinical utility and accuracy of these correction formulas have been increasingly questioned, particularly in hospitalized or critically ill patients.
Several problems are associated with the Payne formula and similar albumin-adjusted calcium calculations:
- Derivation limitations: The original formula was derived from a specific patient cohort and used laboratory methodologies that differ from current routine practices.
- Misclassification: Evidence suggests that these formulas can misclassify a patient's calcium status, performing less effectively than simply evaluating uncorrected total calcium. Studies have shown that in patients with albumin <30 g/L, a significant percentage classified as normocalcemic using the Payne formula actually had hypocalcemia based on ionized calcium levels.
- Inaccuracy in specific populations: The formulas tend to overestimate calcium in patients with renal failure, as uremia can affect albumin measurement. In hypoalbuminemic states, the binding constant between albumin and calcium changes, meaning more calcium binds to each available gram of albumin, contrary to the assumptions of the formulas.
- Poor performance in renal impairment: The agreement between adjusted calcium and ionized calcium is even worse for patients with renal impairment.
- Impact of hypoalbuminemia: The poorer the performance of the adjustment formula, the more severe the hypoalbuminemia.

Payne himself acknowledged that his original formula is not universally applicable and may require modification for specific laboratory assays. Furthermore, any albumin-based adjustment is likely to overestimate calcium in patients with renal failure.
When to Measure Ionized Calcium
Direct measurement of ionized calcium is recommended when an accurate assessment is critical. This includes situations such as:
- Critical illness
- Renal failure
- Abnormalities in calcium-binding proteins
- When results of a total calcium test are not normal and a more precise measurement is needed
- Suspected parathyroid disorders
- During major surgery or when receiving blood transfusions
- Acute pancreatitis symptoms
- Pregnancy with neonatal monitoring
Ionized calcium tests require prompt analysis of blood samples after collection due to the influence of pH and other pre-analytical factors that can affect results if the sample is not handled correctly. Fluctuations in serum pH significantly impact calcium measurements; alkalosis (increased serum pH) leads to increased protein binding and decreased ionized calcium, while acidosis (low pH) increases ionized calcium levels.
Interpreting Calcium Levels: Hypocalcemia and Hypercalcemia
Abnormal calcium levels can manifest in various ways, impacting multiple organ systems.
Hypocalcemia (Low Calcium Levels)
Hypocalcemia can be caused by dietary deficiencies, disorders of the parathyroid axis (e.g., after surgery or autoimmune destruction of parathyroid glands), vitamin D deficiency, malabsorption, or kidney problems. Symptoms may include muscle cramps, numbness or tingling in extremities, fatigue, confusion, memory loss, and irregular heart rhythms. Severe hypocalcemia can lead to seizures or cardiac arrhythmias.
Normal Ionized Calcium Ranges (may vary by laboratory):
| Age Range | Ionized Calcium Normal Range (mg/dL) |
|---|---|
| Newborns (0 to 2 months) | 4.2 to 5.58 |
| Children (2 months to 18 years) | 4.8 to 5.52 |
| Adults (18+ years) | 4.5 to 5.6 |
Treatment for low ionized calcium may involve calcium and vitamin D supplements, calcium gluconate injections, or parathyroid hormone replacement, alongside addressing any underlying conditions.
Hypercalcemia (High Calcium Levels)
Hypercalcemia is commonly associated with dehydration, primary hyperparathyroidism, and malignancy (such as multiple myeloma). Mild to moderate hypercalcemia (total calcium 2.5-3.5 mmol/L or ionized 1.3-2.0 mmol/L) may not require immediate therapy but warrants close monitoring. Severe hypercalcemia (total calcium >3.5 mmol/L or ionized >2.0 mmol/L) requires immediate intervention.
Symptoms of hypercalcemia can include excessive thirst, frequent urination, abdominal pain, bone pain, muscle weakness, fatigue, confusion, nausea, vomiting, constipation, and in severe cases, cardiac arrhythmias or coma. The most common cause of high blood calcium levels is primary hyperparathyroidism, accounting for over 90% of cases.
Treatment for high ionized calcium depends on the cause and may include calcimimetics, calcitonin, bisphosphonates, denosumab, prednisone, IV fluids, or parathyroidectomy (surgical removal of abnormal parathyroid tissue).
Hypercalcemia - Too Much Calcium, Animation
Factors Affecting Calcium Measurements
Several factors can influence calcium measurements, leading to potential errors:
- Venous occlusion: Prolonged tourniquet application during venipuncture can increase plasma protein concentration, falsely elevating total serum calcium.
- Posture: Standing up from a supine position can cause a slight increase in serum calcium.
- Hemolysis: Red blood cells may take up calcium after prolonged contact with the serum, affecting results.
- Anticoagulants: Anticoagulants like EDTA, citrate, and oxalate chelate calcium, rendering it unmeasurable.
- pH fluctuations: As mentioned, pH changes in the blood or sample can alter protein binding and thus ionized calcium levels.
- Diurnal variation: Calcium levels can exhibit mild diurnal variation, with peak levels typically occurring in the late evening.
- Diet: Daily calcium intake can contribute to serum calcium concentration, so fasting before a test may be recommended in some cases.
Given these potential interferences and the limitations of albumin-adjusted formulas, direct measurement of ionized calcium is often preferred for the most accurate assessment of calcium status, particularly in complex clinical scenarios.