Demodex blepharitis is a common yet frequently overlooked eye condition stemming from microscopic Demodex mites that inhabit the eyelids. These tiny creatures can obstruct the oil glands in the eyelids, triggering inflammation, irritation, and the accumulation of crusty debris, known as collarettes, at the base of the eyelashes. If left unaddressed, Demodex blepharitis can significantly diminish one's quality of life through persistent eyelid discomfort and potential vision impairment.
Xdemvy (lotilaner ophthalmic solution 0.25%) has been developed as a targeted treatment to manage this condition. Its primary goal is to eradicate the Demodex mites, alleviate inflammation, and clear the crusting that characterizes Demodex blepharitis. Notably, Xdemvy is the only FDA-approved medication specifically indicated for the treatment of Demodex blepharitis.
Mechanism of Action
The active ingredient in Xdemvy is lotilaner, an antiparasitic agent designed to target Demodex mites. Lotilaner functions by inhibiting specific proteins within the mites' nervous systems, leading to their paralysis and subsequent death. These targeted proteins, identified as GABA-gated chloride channels, are unique to Demodex mites and do not adversely affect the human eye when the medication is used as directed. By effectively eliminating these mites, Xdemvy helps to reduce the inflammation and debris contributing to the symptoms of Demodex blepharitis.
How to Use Xdemvy
It is crucial to use Xdemvy precisely as prescribed by your healthcare provider. The recommended dosage and treatment duration are as follows:
- Dose: Apply one drop into each eye twice daily, with approximately 12 hours between applications.
- Treatment Duration: Continue the treatment regimen for a total of six weeks.
Important Administration Instructions:
- Contact Lenses: If you wear contact lenses, remove them before applying Xdemvy. Wait for at least 15 minutes after application before reinserting your lenses.
- Multiple Eye Drops: If you are using other eye medications concurrently, ensure a minimum five-minute interval between the application of each product.
- Hygiene: Always wash your hands thoroughly before and after applying Xdemvy.
- Sterility: To prevent contamination, avoid touching the tip of the bottle to your eye or any other surface.
- Missed Dose: If you miss a dose, apply it as soon as you remember. However, if it is nearly time for your next scheduled dose, skip the missed dose and resume your regular dosing schedule. Do not double the dose to compensate for a missed one.
Clinical Studies and Efficacy
The effectiveness and safety of Xdemvy were rigorously evaluated in two pivotal clinical studies: Saturn-1 and Saturn-2. These randomized, double-blind studies involved a total of 833 participants diagnosed with Demodex blepharitis. In these studies, participants were unaware whether they were receiving Xdemvy or a placebo.
To be eligible for enrollment, participants had to exhibit specific criteria in at least one eye, including:
- Signs of Demodex infestation with more than 10 upper eyelashes showing waxy or scaly buildup.
- At least mild redness along the edge of the upper eyelid.
- An average of 1.5 or more Demodex mites per eyelash upon examination of both upper and lower lids.
Study Demographics:
Saturn-1 included 421 participants with an average age of approximately 67 years; 57% were female. The racial composition was predominantly White (90.7%), followed by Black or African American (6.4%), Hispanic or Latino (5.9%), and Asian (1.2%). Other racial groups comprised less than 1% of participants.
Saturn-2 involved 412 participants with an average age of around 64 years; 48.5% were female. The racial breakdown was similar, with the majority being White (88.1%), followed by Black or African American (8.5%), Hispanic or Latino (8.2%), and Asian (1.4%). Other racial groups accounted for less than 1%.
Key Treatment Outcomes:
The primary measure of success in both studies was the improvement in eyelid condition, specifically the reduction of collarettes (defined as grade 0, or complete collarette cure, meaning no more than two collarettes on each upper eyelid). After six weeks of treatment:
- Participants using Xdemvy demonstrated significantly clearer eyelids, fewer mites, and reduced redness compared to the placebo group.
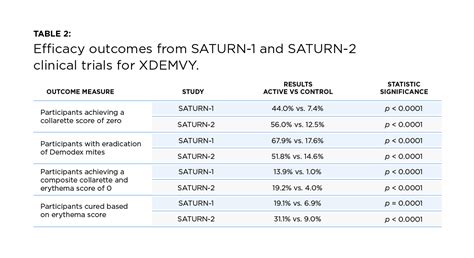
- In Saturn-1, 81.3% of participants in the Xdemvy group experienced a meaningful improvement in collarettes (defined as 10 or fewer, or grade 0 or 1), compared to only 23% in the placebo group. A complete cure (grade 0) was achieved by 44% of Xdemvy users versus 7.4% of placebo users by day 43.
- In Saturn-2, 89.1% of participants in the Xdemvy group showed a clinically meaningful reduction in collarettes, versus 33% in the placebo group. By day 43, 56% of patients in the Xdemvy group achieved a complete collarette cure (grade 0) compared to 12.5% in the placebo group.
Xdemvy was generally well-tolerated in both studies. The most frequently reported side effect was mild stinging upon application.
It is important to note that individual results may vary and may not replicate the outcomes observed in clinical studies.
Preventing Recurrence and Managing Eyelid Health
Implementing simple lifestyle adjustments can aid in preventing the recurrence of blepharitis:
- Maintain consistent eyelid hygiene.
- Always remove eye makeup before sleeping.
- Avoid applying eyeliner to the inner rim of the eyelids, behind the lashes.
- During the initial stages of treatment, refrain from using makeup to prevent further irritation.
- When resuming makeup use, replace all eye cosmetic products, as they may harbor contamination.
- Incorporate omega-3 rich foods into your diet or consider omega-3 supplements.
- For dry eye symptoms, artificial tears can offer relief. However, consult your doctor if frequent or prolonged use is necessary, as over-the-counter drops are not intended for continuous application.
Accessing Xdemvy
Xdemvy is classified as a "specialty" medication. This designation means it is exclusively available through a specialty pharmacy and may necessitate prior authorization from your insurance provider. The drug manufacturer offers assistance programs that could potentially reduce your out-of-pocket costs to as little as $0, depending on your prescription insurance coverage and type.
References:
- XDEMVY. Prescribing Information. Tarsus Pharmaceuticals Inc; 2023.
- Fromstein SR, Harthan JS, Patel J, Opitz DL. Demodex blepharitis: clinical perspectives. Clin Optom (Auckl). 2018;10:57-63.
- Sędzikowska A, Osęka M, Grytner-Zięcina B. Ocular symptoms reported by patients infested with Demodex mites. Acta Parasitol. 2016;61(4):808-814.
- Trattler W, Karpecki P, Rapoport Y, et al. The prevalence of Demodex blepharitis in US eye care clinic patients as determined by collarettes: a pathognomonic sign. Clin Ophthalmol. 2022;16:1153-1164.
- Rhee MK, Yeu E, Barnett M, et al. Demodex blepharitis: a comprehensive review of the disease, current management, and emerging therapies. Eye Contact Lens.