Daewoong Pharmaceutical is spearheading the development of an innovative obesity treatment utilizing a microneedle patch loaded with glucagon-like peptide-1 (GLP-1) analogues. This advanced formulation, featuring an ultra-small 1cm² patch applied to the arm or abdomen, promises significantly enhanced user convenience compared to traditional obesity treatments. The company plans to initiate Phase 1 clinical trials early next year, with the ambitious goal of commercializing the product by 2028.
The company is developing an innovative, painless microneedle obesity treatment that can be applied weekly to the arms or abdomen, delivering medication directly through the microvasculature. This offers a convenient alternative to traditional, injectable GLP-1 analogue therapies. It maintains the efficacy of existing injectables and offers the significant benefit of room temperature storage, eliminating the need for a cold storage system. This represents a marked improvement over oral obesity treatments, which suffer from low absorption rates of about 1% bioavailability and often cause unpleasant side effects such as vomiting, nausea, and diarrhoea.
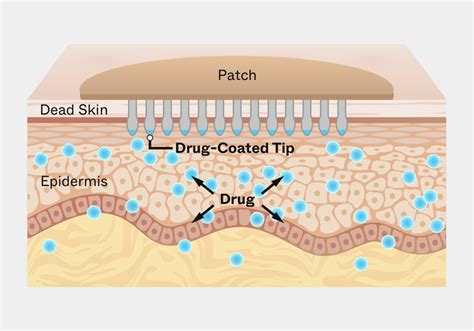
Advancements in Microneedle Technology
Daewoong Therapeutics, a subsidiary of Daewoong Pharmaceutical, has successfully completed preclinical studies and compiled data for a microneedle patch within the Semaglutide series, a GLP-1 analogue. This development leverages their proprietary Closed-Packed Aeroperssured Microneedle (CLOPAM) platform. This platform is protected by six international and 23 domestic patents, with five already registered, covering various aspects of its manufacturing and packaging. The CLOPAM technology addresses previous challenges in drug delivery, significantly enhancing drug uniformity, preventing contamination, and ensuring stability. The microneedle manufacturing process preserves the integrity of the biodegradable needles, which are sensitive to moisture and impact, until their actual use. These soluble microneedles release the drug into the body as they dissolve.
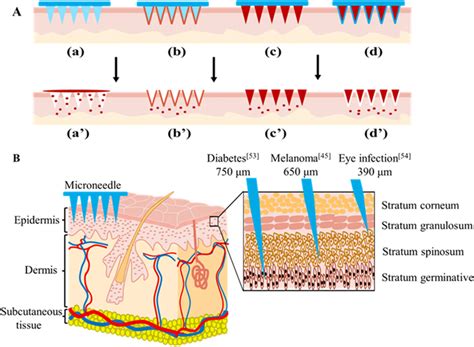
Market Potential and Commercialization Goals
Industry forecasts from Morgan Stanley predict a substantial expansion in the global obesity treatment market, with expectations to reach 104 trillion won by 2030. Daewoong Pharmaceutical is strategically positioned to enter this burgeoning market with its innovative microneedle obesity treatment by 2028. The company aims to revolutionize the sector by effectively addressing the limitations of current therapies, particularly in improving treatment adherence.
Understanding GLP-1 Medications and Their Administration
Glucagon-like peptide-1 (GLP-1) receptor agonists are FDA-approved medications primarily utilized for managing type 2 diabetes and, in specific formulations, for weight management in adults with a BMI of ≥30 kg/m² or ≥27 kg/m² with weight-related comorbidities. While most GLP-1 medications are administered via subcutaneous injection (e.g., semaglutide, liraglutide, dulaglutide, or tirzepatide), with oral semaglutide (Rybelsus) being the only FDA-approved non-injectable option, transdermal patch delivery systems represent an emerging area of pharmaceutical development.
It is crucial to note that, as of current FDA approvals, there are no commercially available GLP-1 patches approved for clinical use in the United States. The concept behind transdermal GLP-1 delivery involves using a patch applied to the skin that would theoretically release medication through the dermal layers into systemic circulation. This mechanism would bypass the gastrointestinal tract and provide steady-state drug levels over an extended period. Research into transdermal delivery of peptide medications faces significant challenges due to the large molecular size of GLP-1 analogs and the skin's natural barrier function. While microneedle patches and other advanced delivery technologies are under investigation in preclinical and early clinical studies, any information about "wearing" GLP-1 patches currently refers to investigational products not yet available for prescription use.
Patients should exercise caution and avoid purchasing unapproved GLP-1 patches marketed online, as these products may be counterfeit or unsafe. Compounded medications and specific branded medications like Zepbound® are available, but compounded medications are prepared by licensed pharmacies and are not FDA-approved. References to brands like Wegovy®, Ozempic®, Rybelsus®, Mounjaro®, or Saxenda®, or other GLP-1 brands, are for informational purposes only. Compounded and FDA-approved medications are not interchangeable.
Instructions for Injectable GLP-1 Medications (for context)
For individuals currently using injectable GLP-1 medications, proper administration is essential. After an initial consultation, patients are instructed on how to use these medications, which are typically delivered at home. The necessary supplies include a vial, a syringe, and an alcohol pad. Once a vial has been opened and exposed to air and potential germs, it is important to maintain sterility. Uncapping the insulin syringe requires care due to the small needle size. Syringes come in various sizes; for example, a 5.0 ml syringe can be used to draw up 2.5 mL of medication.
Before drawing any liquid, hold the needle perpendicular to the bottle and insert it straight into the center of the vial. Once inside, invert the vial. Holding the vial securely with one hand allows the other hand to draw the liquid into the syringe. If air is present in the syringe, gently tap it with a finger to dislodge the air bubbles, being careful not to bend the needle. It may be necessary to pull back slightly more and then push back up to the desired measurement marker. The needle can then be either laid on the alcohol pad or recapped, though recapping is often avoided.
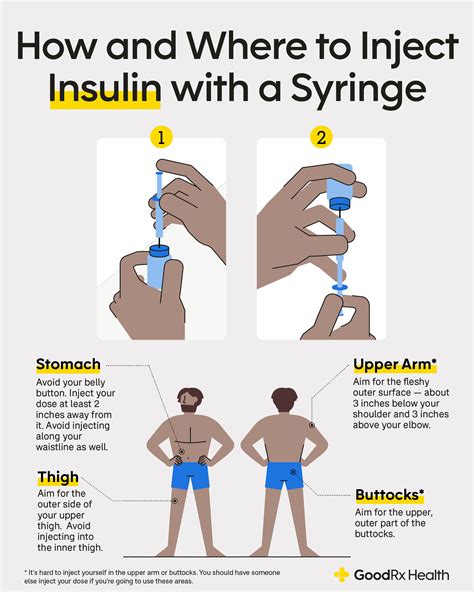
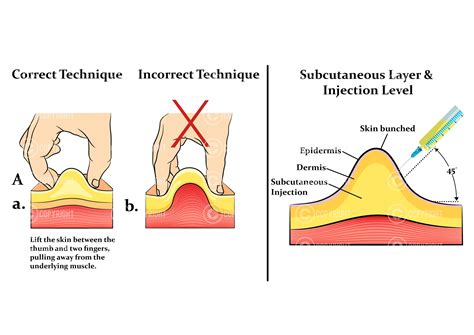
Subcutaneous Injection Technique
These injections are subcutaneous (SQ) injections, meaning they must be delivered into the fatty tissue. Common injection sites include the abdomen, approximately two inches away from the belly button, or the back part of the arm if someone else is administering the injection. Injections can also be given in the leg. It is critical that the medication enters the SQ tissue; if it enters the muscle, its effectiveness will be reduced.
To administer the injection, uncap the needle. If self-administering, pinch up a small amount of skin between your fingers. The needle should be inserted straight in, perpendicular to the skin, without any angle, due to its small size. Avoid having your finger on the plunger while inserting the needle to prevent accidental medication delivery before the needle is fully inserted. Push the needle straight in, allow a second or two for all the medicine to be delivered, and then pull the needle straight back out. The used needle should then be disposed of in a proper sharps container.
Typically, bruising or bleeding after injections should not occur, although it can happen occasionally. Mild redness or irritation at the injection site is possible, which is why rotating injection sites is recommended.
Optimal Body Sites for Transdermal Application
While no GLP-1 patches have established application sites due to their investigational status, general principles of transdermal drug delivery offer guidance. Common sites for transdermal medication delivery include the upper outer arm, upper chest, upper back between the shoulder blades, and the abdomen (avoiding the waistline). These areas typically provide sufficient skin surface area, consistent drug absorption, and are accessible for application while remaining concealed under clothing. Site selection considers factors such as skin thickness, local blood flow, hair density, and the potential for patch dislodgement.
Site rotation is a fundamental principle in transdermal therapy to minimize local skin irritation and prevent contact dermatitis. Patients should avoid applying a new patch to the exact same location for at least one to two weeks, or as specified by product labeling. For injectable GLP-1 medications, recommended sites include the abdomen (at least 2 inches from the navel), the front of the thighs, and the upper arms. Rotating these sites with each injection helps prevent lipodystrophy (changes in subcutaneous fat tissue that can affect medication absorption). Patients must never inject into areas with lipohypertrophy, scars, bruises, or skin abnormalities. The abdomen generally offers the most consistent absorption for subcutaneous injections, though all approved sites are clinically effective with proper technique.
Common Mistakes to Avoid
Even though GLP-1 patches are not yet FDA-approved, understanding common errors associated with transdermal medication use is beneficial. A frequent mistake is inadequate skin preparation, which can compromise drug absorption and patch adhesion. Applying patches to inappropriate body sites-such as areas with excessive hair, skin folds, or locations subject to frequent movement or friction-is another critical error, increasing the likelihood of patch displacement and subtherapeutic drug levels.
Exposing transdermal patches to external heat sources (heating pads, saunas, hot tubs, prolonged sun exposure) can significantly increase drug absorption and potentially lead to overdose. Patients should also avoid wearing multiple patches simultaneously unless specifically directed by their healthcare provider. Failure to adequately rotate application sites is another common mistake, which can cause contact dermatitis, skin sensitization, or localized tissue changes that impair drug absorption.
For patients using currently available injectable GLP-1 medications, common errors include injecting into the same site repeatedly, failing to allow refrigerated medication to reach room temperature before injection, not rotating injection sites properly, and incorrect pen device technique. Injecting cold medication can increase discomfort, while poor site rotation may lead to lipohypertrophy or lipoatrophy. Patients should never share pens, must use a new needle for each injection, and should not store pens with needles attached. Proper disposal of used needles in FDA-cleared sharps containers is essential for safety.
Patch Replacement and Expected Outcomes
For any transdermal medication system, the prescribed wear time is determined by the drug's pharmacokinetic profile and the patch's delivery technology. Transdermal patches typically remain in place for periods ranging from 24 hours to seven days. Since no GLP-1 patches are currently FDA-approved, there is no established wear schedule for such products.
When changing any transdermal patch, patients should remove the old patch carefully, fold it in half with the adhesive sides together, and dispose of it safely according to the medication guide. The new patch should be applied to a different site, following the principles of rotation. During the initial period of using any new transdermal medication, patients may experience local skin reactions such as mild redness, itching, or irritation at the application site. These reactions are often transient and resolve within a few hours after patch removal.
For injectable GLP-1 medications, common side effects include nausea, vomiting, diarrhea, and decreased appetite, particularly during dose escalation. These gastrointestinal effects typically diminish over time. More serious potential adverse effects include pancreatitis, gallbladder disease, acute kidney injury, and an increased risk of hypoglycemia when used with insulin or sulfonylureas. Patients should contact their healthcare provider if they experience severe abdominal pain, persistent vomiting, signs of pancreatitis, symptoms of gallbladder disease, or symptoms of thyroid tumors.
Frequently Asked Questions
Are GLP-1 patches available for purchase in the United States?
No, there are currently no FDA-approved GLP-1 patches available for clinical use in the United States. All approved GLP-1 medications require subcutaneous injection or oral administration (Rybelsus). Patients should avoid purchasing unapproved patches marketed online.
What are the approved methods for taking GLP-1 medications?
FDA-approved GLP-1 medications are administered via subcutaneous injection into the abdomen, thigh, or upper arm, or taken orally as Rybelsus. Proper injection technique includes rotating sites and using a new needle for each injection.
Why aren't GLP-1 patches available yet?
Transdermal delivery of GLP-1 medications faces significant challenges due to the large molecular size of GLP-1 analogs and the skin's natural barrier function. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are among the most effective treatments for obesity. The current therapy associated with repeated subcutaneous injections to maintain drug efficacy leads to patient compliance issues and raises environmental concerns due to sharp biohazard waste from disposed syringes/needles. Research is ongoing to develop advanced delivery systems, such as programmable scheduled release microneedles (PSR-MNs), which aim to enable repeated drug release over extended periods, simulating multiple injections with a single patch application.
How to Inject Your Semaglutide/Tirzepatide (GLP-1 Analogs)
tags: #glp #1 #nano #microneedle #patch #instructions